Are alkenes polar?

Sarah Smith
Published Jan 22, 2026
Alkenes are nonpolar, since they contain nothing but carbon-carbon and carbon-hydrogen bonds, and are not soluble in water; they are also generally less dense than water.
Do alkenes have polar bonds?
I think it should give some polarity because C is slightly more electronegative than H, so the electrons in the C–H bond will be slightly polarised towards C. But alkenes are typically referred to as non-polar.
Are alkenes and alkanes polar?
There is not a significant difference in electronegativity between C and H atoms, so these are not polar molecules.
Are alkynes polar?
Alkynes are nonpolar, since they contain nothing but carbon and hydrogen, and so, like the alkanes and alkenes, they are not soluble in water, and are generally less dense than water.
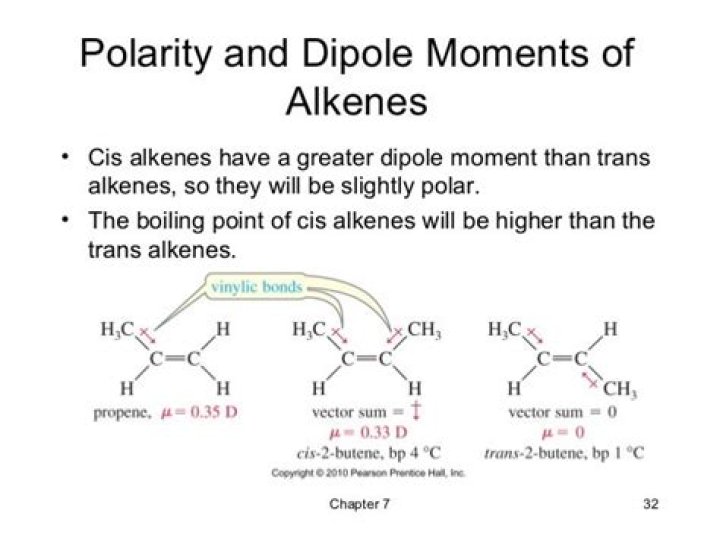
Why CIS alkenes are polar?
In cis isomer two polar groups attached to the same side of a restricted system and form an unsymmetrical molecule. Each bond have dipole movement and both the bond produce a resultant dipole moment in one direction, so cis form of alkene are polar.
23 related questions foundAre alkenes odorless?
Alkenes are colourless and odourless with an exception of ethene which is a colourless gas with pleasant smell.
Are alkenes reactive?
Alkenes are relatively stable compounds, but are more reactive than alkanes because of the reactivity of the carbon–carbon π-bond. Most reactions of alkenes involve additions to this π bond, forming new single bonds.
Are alkenes and alkynes soluble in water?
Alkanes and alkenes are non-polar molecules. Water, on the other hand, is a polar molecule. So, alkanes and alkenes are not soluble in water. Alkynes are generally nonpolar molecules with little solubility in polar solvents, such as water.
What are the physical properties of alkenes and alkynes?
Alkynes are nonpolar, unsaturated hydrocarbons with physical properties similar to alkanes and alkenes. Alkynes dissolve in organic solvents, have slight solubility in polar solvents, and are insoluble in water. Compared to alkanes and alkenes, alkynes have slightly higher boiling points.
Why alkanes alkenes and alkynes are non polar compounds?
Carbon has an electronegativity of 2.5 while Hydrogen has an electronegativity of 2.2. This is a difference of only . 3 electronegativity. This makes the compound non polar.
Are alkenes volatile?
Longer-chain alkenes are less volatile and they have higher boiling points and melting points. Longer-chain alkenes have higher density and viscosity values.
What intermolecular forces do alkenes have?
The strongest intermolecular forces between alkene molecules are Van der Waals' forces (remember him?). And the longer the alkene chain, the higher the boiling point becomes. Lastly, let's talk about p orbitals. These are needed to form pi bonds (covalent chemical bonds).
Is alkane polar or non polar?
Alkanes are nonpolar molecules, since they contain only nonpolar carbon-carbon and carbon-hydrogen bonds.
Are alkanes and alkenes both nonpolar?
Similarities Between Alkanes and Alkenes
The atoms in alkanes and alkenes are bonded to each other through covalent bonds. Both are nonpolar molecules. Therefore both are insoluble in water and other polar solvents.
What are the properties of alkenes?
Physical Properties of Alkenes
- Physical State. These double-bonded compounds are colourless and odourless in nature. ...
- Solubility. The alkenes are insoluble in water due to their nonpolar characteristics. ...
- Boiling Point. ...
- Melting Point. ...
- Polarity.
Why are alkynes slightly polar?
Now Alkynes are slightly electronegative in nature. The triply bonded carbon atoms in alkynes are sp hybridized, Whereas like in alkanes the single bond atoms are sp3 hybridized, causing the difference in the electronegativity. This makes it easier for them to attract the shared electron pair of the C-H bond.
Are alkynes hydrophobic or hydrophilic?
Our findings suggest that the triple bonded carbons in 2-alkyne that reduce hydration water act as a hydrophobic group in 2-alkyne. Thus, the methylene group should be called “hydrophilic” in this case because it actually recovers the hydration water when placed next to more hydrophobic groups.
Why are alkenes more soluble than alkanes?
Alkenes are more soluble in water than alkanes because the carbon to carbon double bond contains a pi bond.
Are alkenes soluble in polar solvent?
Density. Alkenes are lighter than water and are insoluble in water due to their non-polar characteristics. Alkenes are only soluble in nonpolar solvents.
Are alkynes soluble in non polar?
Alkynes are generally nonpolar molecules with little solubility in polar solvents, such as water. Solubility in nonpolar solvents, such as ether and acetone, is extensive.
Are alkynes reactive?
Thus, alkynes are very reactive, and with the exception of ethyne, commonly referred to as acetylene (C2H2), they are not commonly encountered. The naming system for alkynes follows the conventions for the alkanes and alkenes, however the presence of the triple bond is indicated by the -yne termination.
Are alkenes neutral?
Neutral compounds include the following functional group classes: hydrocarbons (alkanes, alkenes, alkynes, aromatic compounds), alcohols, aldehydes, ketones, esters, amides, nitro compounds.
How do you know if alkenes are stable?
The relative stability of alkenes is determined by comparing their heats of hydrogenation. The smaller the heat of hydrogenation, the higher the stability of the alkene.
Why are alkenes less stable than alkanes?
Alkanes have a single bond, less energy than alkenes and alkynes which have respectively two and three bonds and higher energy. Higher energy means shorter bonds which means stronger bonds. But in this case, the stronger bonds in alkenes/alkynes have higher bond energy and thus more unstable than alkanes.