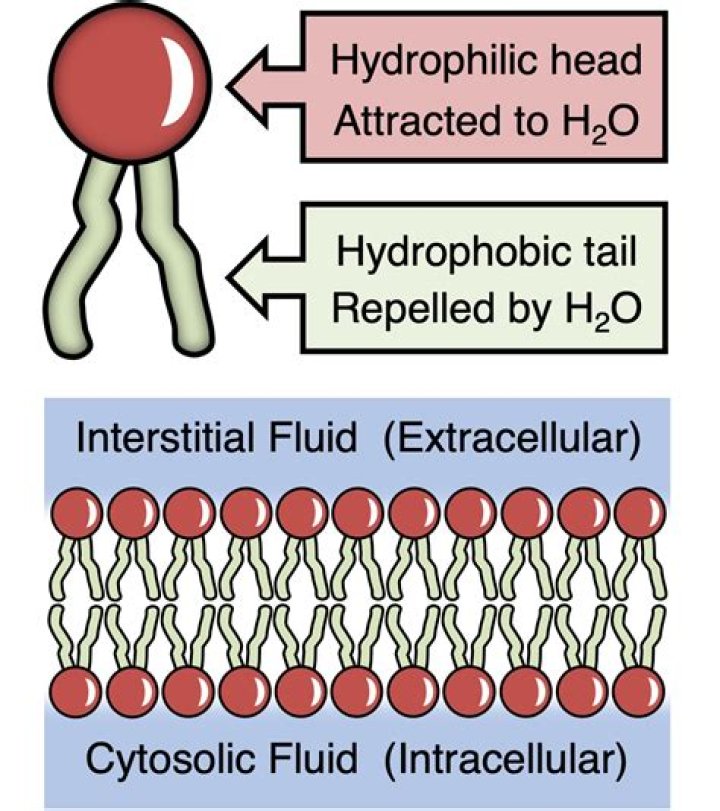
Are alkynes hydrophobic or hydrophilic?

Emma Payne
Published Jan 11, 2026
Our findings suggest that the triple bonded carbons in 2-alkyne that reduce hydration water act as a hydrophobic group in 2-alkyne. Thus, the methylene group should be called “hydrophilic” in this case because it actually recovers the hydration water when placed next to more hydrophobic groups.
Are alkanes hydrophilic or hydrophobic?
Although short n-alkane chains are classic examples of hydrophobic solutes, mounting evidence points to a hydrophilic crossover for the hydration free energies (ΔG) of sufficiently long n-alkane chains.
Why are alkenes called hydrophobic?
Hydrocarbon chains are non-polar and do not form bonds with water molecules. They are said to be hydrophobic (water hating). Some functional groups form hydrogen bonds with water, allowing the molecule to dissolve.
Are alkynes non-polar?
Because of this linear shape, geometric isomerism does not occur in alkynes. Alkynes are nonpolar, since they contain nothing but carbon and hydrogen, and so, like the alkanes and alkenes, they are not soluble in water, and are generally less dense than water.
Does alkyne dissolve in water?
Alkynes are generally nonpolar molecules with little solubility in polar solvents, such as water.
42 related questions foundWhat are the physical properties of alkynes?
Alkynes are nonpolar, unsaturated hydrocarbons with physical properties similar to alkanes and alkenes. Alkynes dissolve in organic solvents, have slight solubility in polar solvents, and are insoluble in water. Compared to alkanes and alkenes, alkynes have slightly higher boiling points.
Are all alkanes soluble or insoluble in water?
Alkanes are not soluble in water, a polar substance. The two substances do not meet the usual criterion of solubility: “Like dissolves like.” Water molecules are too strongly attracted to each other by hydrogen bonds to allow nonpolar alkanes to slip in between them and dissolve.
Which alkane is least soluble in water?
The answer is d) 1-pentanol.
What are alkanes alkenes and alkynes?
Alkanes have only single bonds between carbon atoms and are called saturated hydrocarbons. Alkenes have at least one carbon-carbon double bond. Alkynes have one or more carbon-carbon triple bonds. Alkenes and alkynes are called as unsaturated hydrocarbons.
Why is pentane insoluble in water?
The pentanes are molecular, and certainly they are non-polar, and hence addition of n-pentane to water gives rise to two immiscible layers, with the LESS dense pentane floating on top of the funnel.
Why are alkynes slightly polar?
Now Alkynes are slightly electronegative in nature. The triply bonded carbon atoms in alkynes are sp hybridized, Whereas like in alkanes the single bond atoms are sp3 hybridized, causing the difference in the electronegativity. This makes it easier for them to attract the shared electron pair of the C-H bond.
Why are alkynes weakly polar?
This is because in alkynes there are 3 bonds between at least two carbons (that is 6 electrons). So the bond enthalpy (energy required to break the bond) is maximum. Alkenes have 2 bonds between at least two carbons. In alkanes all are single bonds.
Are alkynes solid liquid or gas?
Alkynes are generally gases, and are soluble in organic solvents, like acetone or benzene, and insoluble in water. Their boiling points and acidities are higher compared to the other hydrocarbons, like alkanes and alkenes.
Why alkynes are slightly soluble in water?
Due to greater electronegativity of C-atom in alkyne, the π-electrons are more tightly held in the triple bond then the π-electrons of double bond in alkene and sigma electrons of alkane. So, the alkyne is more polar than alkene and alkane, and hence is slightly more soluble in water ,which is a polar solvent.
What is the solubility of alkynes?
The solubility of Alkynes
Alkynes are generally nonpolar molecules with little solubility in polar solvents, such as water. Insoluble in water. Soluble in various organic solvent. They are insoluble in water but readily dissolve in organic solvents such as ether, carbon tetrachloride, and benzene.
Are alkynes linear?
The linear or straight geometry is a primary characteristic of alkynes with a carbon-carbon triple bond. A part of the molecule is in a single-dimensional straight line. Ethyne is used to make a variety of other compounds.
Why alkynes have slight solubility in polar solvents?
Due to greater electronegativity of C-atom in alkyne, the π-electrons are more tightly held in the triple bond then the π-electrons of double bond in alkene and sigma electrons of alkane. So, the alkyne is more polar than alkene and alkane, and hence is slightly more soluble in water ,which is a polar solvent.
What intermolecular forces are present in alkynes?
Importance of Triple Bonds
Hybridization due to triple bonds allows the uniqueness of alkyne structure. This triple bond contributes to the nonpolar bonding strength, linear, and the acidity of alkynes. Physical Properties include nonpolar due to slight solubility in polar solvents and insoluble in water.
Are alkynes reactive?
Thus, alkynes are very reactive, and with the exception of ethyne, commonly referred to as acetylene (C2H2), they are not commonly encountered. The naming system for alkynes follows the conventions for the alkanes and alkenes, however the presence of the triple bond is indicated by the -yne termination.
What are the chemical reactions of alkynes?
The principal reaction of the alkynes is addition across the triple bond to form alkanes. These addition reactions are analogous to those of the alkenes. Hydrogenation. Alkynes undergo catalytic hydrogenation with the same catalysts used in alkene hydrogenation: platinum, palladium, nickel, and rhodium.
Do alkynes rearrange?
This intermediate carbocation may rearrange or simply pick up a nucleophile to form the expected addition product. The situation with alkynes is different. Similar protonation of the π bond of an alkyne would produce a very unstable vinyl cation so this does not happen in most cases.
Is pentane least soluble in water?
Pentane is a colorless liquid hydrocarbon, with a characteristic smell. It is insoluble in water, but miscible with other organic solvents.
How soluble is pentane in water?
Pentane has a molecular weight of 72.15 g mol−1. At 25 °C, pentane has a solubility in water of 38 mg l−1, an estimated vapor pressure of 514 mm Hg, and a Henry's law constant of 1.25 atm-m3 mol−1 ().