Can alkenes be oxidised by potassium dichromate?

Daniel Moore
Published Jan 18, 2026
Oxidation of Alkenes
Can alkenes be oxidised?
Alkenes can easily be oxidized by potassium permanganate and other oxidizing agents. What products form depend on the reaction conditions. At cold temperatures with low concentrations of oxidizing reagents, alkenes tend to form glycols. This reaction is sometimes referred to as the Baeyer test.
What can be oxidized by K2Cr2O7?
Description: Primary and secondary alcohols are oxidized by K2Cr2O7 to carboxylic acids and ketones respectively. The oxidation is physically observed by the change in color upon reduction of Cr6+ (yellow) to Cr3+ (blue).
Does potassium permanganate oxidize alkenes?
The acidified potassium manganate(VII) solution oxidizes the alkene by breaking the carbon-carbon double bond and replacing it with two carbon-oxygen double bonds. The products are known as carbonyl compounds because they contain the carbonyl group, C=O.
Can you oxidize alkanes?
Alkanes can be oxidized to carbon dioxide and water via a free‐radical mechanism. The energy released when an alkane is completely oxidized is called the heat of combustion.
41 related questions foundIs alkene to alkane reduction or oxidation?
One important alkene addition reaction is hydrogenation., where the alkene undergoes reduction to an alkane. In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane.
Are alkenes more oxidized than alkanes?
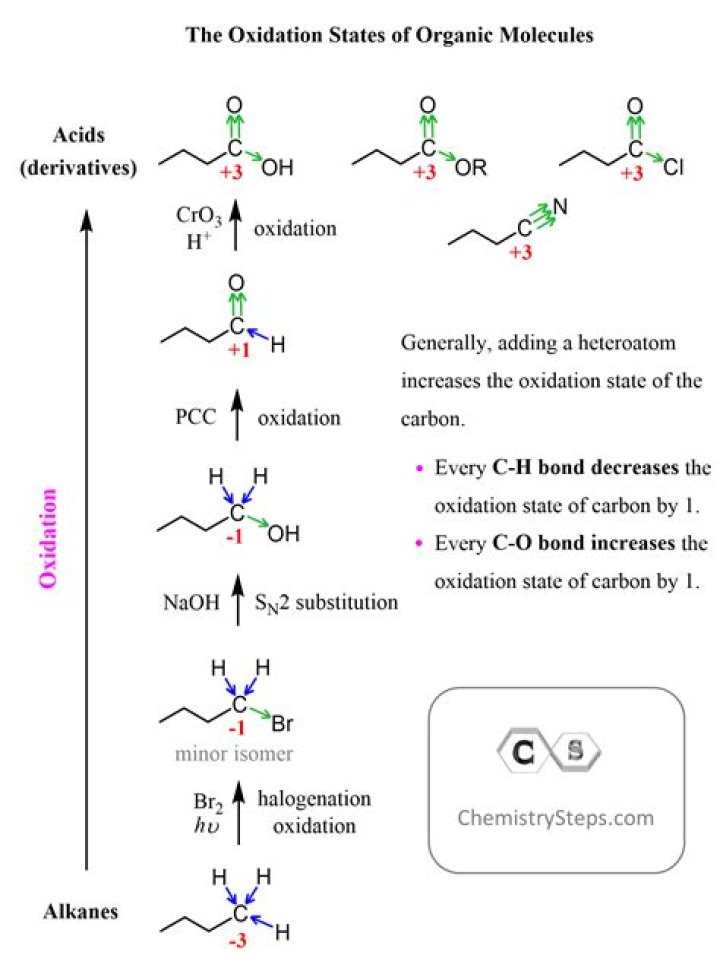
Alkanes are highly reduced, while alcohols - as well as alkenes, ethers, amines, sulfides, and phosphate esters - are one step up on the oxidation scale, followed by aldehydes/ketones/imines and epoxides, and finally by carboxylic acid derivatives (carbon dioxide, at the top of the oxidation list, is specific to the ...
How does potassium permanganate oxidize?
Potassium permanganate oxidizes aldehydes to carboxylic acids, such as the conversion of n-heptanal to heptanoic acid: 5 C6H13CHO + 2 KMnO4 + 3 H2SO4 → 5 C6H13COOH + 3 H2O + K2SO4 + 2 MnSO. Even an alkyl group (with a benzylic hydrogen) on an aromatic ring is oxidized, e.g. toluene to benzoic acid.
What happens to alkenes when they are oxidized by KMnO4?
When a purple solution of the oxidizing agent KMnO4 is added to an alkene, the alkene is oxidized to a diol and the KMnO4 is converted to brown MnO2. Thus, if the purple color changes to brown in this reaction, it is a positive reaction. The diol produced has two adjacent alcohol groups.
Is potassium dichromate an oxidising agent?
Potassium dichromate is an oxidising agent in organic chemistry, and is milder than potassium permanganate. It is used to oxidize alcohols. It converts primary alcohols into aldehydes and, under more forcing conditions, into carboxylic acids.
What product is formed when 1 propanol is oxidized by using potassium dichromate K2Cr2O7 in an aqueous acid solution?
Hello Dear. When 1- propanol undergoes oxidation with acidified K₂Cr₂O₇ , It gives Aldehyde and then further formed carboxylic acid. Primary alcohal --→ Aldehyde + water --→ Carboxylic acid + H⁺ .
Why is potassium dichromate a good oxidizing agent?
Potassium dichromate is a good oxidizing agent because when elements come into its contact in a chemical reaction they become more electronegative as their atom's oxidation state increases.
Can cyclohexanol be oxidized?
Primary alcohols may be oxidized to aldehydes, and then to carboxylic acids. Secondary alcohols may be oxidized to ketones, while tertiary alcohols will not oxidize. This exercise will oxidize cyclohexanol to cyclohexanone using and acidic solution of sodium hypochlorite.
Why is potassium manganate colorless?
The solution will gradually become colourless as the sulfur dioxide reacts with the potassium permanganate. The colour of the manganate ion is lost as the sulfurous acid is oxidized to sulfuric acid.
Do alkanes react with potassium permanganate?
Hydrocarbons with only single bonds are called alkanes. Alkanes are called saturated hydrocarbons because each carbon is bonded with as many hydrogen atoms as possible. Potassium permanganate will not react with alkanes since they are saturated.
Is potassium manganate an oxidising agent?
Potassium manganate(VII), KMnO4, is a deeply coloured purple crystalline solid. It is a powerful oxidising agent. In acidic solution, it undergoes a redox reaction with ethanedioate ions, C2O42-. The MnO4- ions are reduced to Mn2+ and the C2O42- ions are oxidised to CO2.
Which of the following Cannot be obtained on oxidation of alkenes with potassium permanganate followed by acid hydrolysis?
Hence aldehydes cannot be obtained when alkenes are oxidized with KMnO4.
Which alkene is on oxidation with acidic KMnO4?
An alkene on vigorous oxidation with `KMnO_(4)` solution produces only acetic acid.
Which of the following compounds Cannot be oxidized by KMnO4?
KmNo4 cannot be oxidised by o3.
What is the oxidation number of potassium in potassium permanganate?
If you're familiar with the rules for calculating oxidation numbers (more here: ), you'll notice immediately that each oxygen atom has an O.N. of -2 and that the potassium atom has an O.N. of +1.
Is permanganate an oxidizing agent?
Potassium permanganate, KMnO4, is a powerful oxidizing agent, and has many uses in organic chemistry.
How does acidic potassium permanganate reacts with ferrous sulphate and so2?
Explanation: When acidified potassium permanganate reacts with ferrous sulphate, it forms potassium sulphate, ferric sulphate, manganese sulphate and water.
What is the product of oxidation of alkene?
The products of the strong oxidation of alkenes can include carbon dioxide, water, alkanones (ketones) and alkanoic acids (carboxylic acids) depending on the location of the double bond within the alkene molecule.
How do you go from alkanes to alkenes?
An alkene represents an unsaturated hydrocarbon with double bonds, while an alkane is a saturated hydrocarbon with only single bonds. To convert an alkane to an alkene, requires that you remove hydrogen from the alkane molecule at extremely high temperatures. This process is known as dehydrogenation.
What functional groups Cannot be oxidized?
For alcohols, both primary and secondary alcohols can be oxidized. Tertiary alcohols, on the other hand, cannot be oxidized. In many oxidation reactions the oxidizing agent is shown above the reaction arrow as [O]. The oxidizing agent can be a metal or another organic molecule.