Can silver be purified by electrolysis?

Sarah Smith
Published Jan 22, 2026
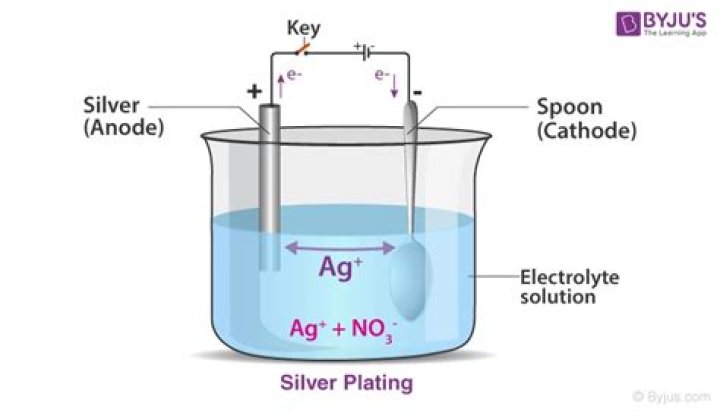
Silver – The electrolytic silver refining process includes a crude silver anode and a refined silver cathode. The electrolytic process is similar to gold, except that the silver anodes are dissolved in a nitric acid bath. The resulting silver will be about 99.9% pure.
How can you purify silver by electrolysis?
Electrorefining involves electrolysis of a silver anode in silver nitrate, low-acid electrolyte to form pure silver crystals on the cathode.
Can silver be refined by electrolysis?
Silver metal can be refined by using the electrolytic refining. When silver is found in ores containing copper then the electrolytic refining method is used to extract the silver in pure form. In this single-displacement reaction, silver nitrate will dissolve copper, producing elemental silver crystals.
Can silver be purified?
Add 3 parts distilled water to 1 part nitric acid. Place on stove or hot plate and simmer until all of the metals are gone. Add 10 parts tap water. Filter off the silver chloride through a plastic funnel or strainer lined with a filter.
How does silver get purified?
Today, the cupellation process involves heating crushed ore on a porous bed of bone ash until it melts. The lead and other impurities are absorbed into the bone ash, and the molten silver is drawn off. Silver is extracted from lead ores in large furnaces, a process called cupellation.
27 related questions foundHow many times is silver purified?
He even uses this image as a picture of His own Word: The words of the LORD are pure words; as silver tried in a furnace on the earth, refined seven times” (Psalm 12:6). Seven is the number of completion, perfection—God's Word is perfect and complete.
Is silver renewable or nonrenewable?
Earth minerals and metal ores like gold, silver, and iron are sometimes also considered to be nonrenewable resources since they're similarly formed from geological processes that span millions of years. On the other hand, renewable resources include solar power, wind power, and sustainably harvested timber.
Can nitric acid dissolve silver?
The full dissolution of silver requires nitric acid, or HNO3, which reacts with silver to form silver nitrate, a water-soluble compound.
How do you refine impure in silver?
Small quantities of silver can be refined by dissolving them in nitric acid. The acid will solubilize the silver and copper and other base metals, leaving any gold or platinum as a sludge on the bottom of the dissolving vessel.
How long does it take nitric acid to dissolve silver?
I'm using 70% Nitric Acid to dissolve Silver (and copper) out of CPU's before further refining. Dropping the silver out of concentration with table salt. It works best when left for 48 hours (although the reaction can be quite violent at first).
How do you purify silver with fire?
As she watched the silversmith working on a particular piece, he explained that the only way to purify and refine silver is to hold it in the middle of the fire where the flames are the hottest so that all the impurities are burned away.
How do you separate silver from other metals?
It is often found in metallic ores that contain quantities of other metals, such as copper. To separate silver from an ore containing a quantity of copper, you need to heat the ore sample to a level sufficient to melt the silver but leave the copper in a still-solid state.
How do you separate silver from lead?
When zinc is added to liquid lead that contains silver as a contaminant, the silver preferentially migrates into the zinc. Because the zinc is immiscible in the lead it remains in a separate layer and is easily removed. The zinc-silver solution is then heated until the zinc vaporizes, leaving nearly pure silver.
What is purification of metal by electrolysis?
The process used in purifying metals by electrolysis is called electrorefining. In the process for refining a metal in an electrolytic cell, in which the impure metal is used as the anode and the refined metal is deposited on the cathode.
How do you refine silver without nitric acid?
Cover the silver with 2 tablespoons of baking soda and 1 tablespoon of salt. Pour the boiling water onto the silver. Only pour enough water into the dish so it's covering the silver. Wait for the water to mix with the baking soda.
Is the refining of silver the recovery of silver from silver nitrate solution involved displacement by copper metal write down the reaction involved?
The recovery of Silver from Silver Nitrate solution involved displacement by Copper metal. Copper (Cu) being a more reactive metal than Silver (Ag), displaces Silver from Silver Nitrate. In this way, silver can be obtained from its solution.
What is the difference between electrolysis and electrolytic refining?
The key difference between electrolytic reduction and refining is that electrolytic reduction method uses graphite electrodes of the same size, whereas electrolytic refining method uses impure metal as the anode and a cathode made of same metal with high purity.
Is it hard to mine silver?
Silver ores
One of the most challenging aspects of silver mining is extraction of the ore, as silver and its host rock can be very heavy. Explosives are frequently used to shatter veins into manageable pieces, which are transported via mine cars and then lifted to the surface.
How do you separate rocks from silver?
When the ore is the cooled down a crust containing silver and zinc forms on the surface. The crust is then collected and then distilled to extract silver from the zinc. When silver is found in ores containing copper then the electrolytic refining method is used to extract the silver.
Does hydrogen peroxide dissolve silver?
The solubility of silver in hydrogen peroxide found by the direct deternlination method indicates that the solubility is a function of peroxide concentration, and does not vary with temperature over the range - 11 to 25°C.
Can aqua regia dissolves silver?
Intriguingly, while aqua regia does dissolve gold, platinum, mercury, and other metals it does not dissolve silver, nor iridium.
What acids refine silver?
Metallic silver can be dissolved from gold alloys of less than 30 percent gold by boiling with 30-percent-strength nitric acid in a process referred to as parting. Boiling with concentrated sulfuric acid to separate silver and gold is called affination.
Can silver be recycled?
Precious metals however, including gold, silver and platinum as well as others are classed as non-ferrous. Non-ferrous metals do not lose their chemical or physical properties in the recycling process, meaning they can be recycled an infinite number of times.
Is silver synthetic or natural?
Silver occurs uncombined, and in ores such as argentite and chlorargyrite (horn silver). However, it is mostly extracted from lead-zinc, copper, gold and copper-nickel ores as a by-product of mining for these metals.
Where does the silver come from?
The metal is found in the Earth's crust in the pure, free elemental form ("native silver"), as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. Most silver is produced as a byproduct of copper, gold, lead, and zinc refining.