Does bromine water react with alkanes?

Mia Kelly
Published Jan 09, 2026
Alkanes are far less reactive than alkenes and will only react with bromine water in the presence of UV light. Under these conditions, alkanes undergo substitution reactions with halogens, and will slowly de-colourise bromine water.
What does bromine water react with?
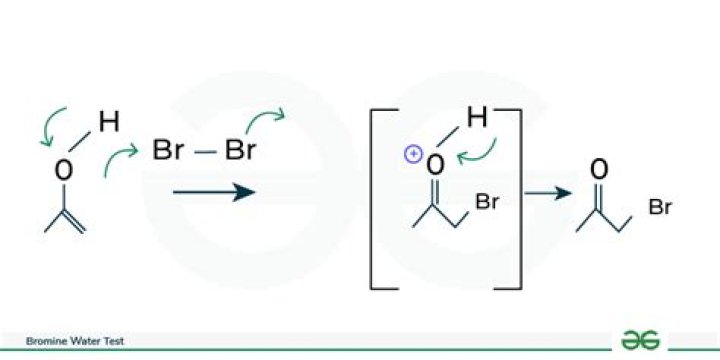
The most common compounds that react well with bromine water are phenols, alkenes, enols, the acetyl group, aniline, and glucose.
What happens when alkane react with bromine?
In the presence of light, or at high temperatures, alkanes react with halogens to form alkyl halides. Reaction with chlorine gives an alkyl chloride. Reaction with bromine gives an alkyl bromide. Unsaturated hydrocarbons such as alkenes and alkynes are much more reactive than the parent alkanes.
How does bromine water test for alkanes?
You can use bromine water, which is an orange solution, to distinguish between alkanes and alkenes. There is no change when bromine water is mixed with an alkane, but it turns colourless when mixed with an alkene.
Does bromine water react with alkyne?
A reaction of bromine with alkenes and alkynes. With alkenes and alkynes, bromine reacts rapidly. This can be used as a visual test to differentiate between alkanes and alkenes and alkynes, which alkanes do not react rapidly with bromine.
24 related questions foundHow can you distinguish between alkanes alkenes and alkynes?
Alkanes have only single bonds between carbon atoms and are called saturated hydrocarbons. Alkenes have at least one carbon-carbon double bond. Alkynes have one or more carbon-carbon triple bonds.
How do alkenes react with bromine water?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot.
Why do alkanes not react with bromine water?
Alkanes do not react with spontaneously bromine water due to their saturated nature. Bromine is non-polar and therefore dissolves more readily in a non-polar alkane than in polar water. Therefore, when alkanes come into contact with bromine water, they cause it to decolourise, while they adopt the colour.
Why do alkanes not react with potassium permanganate?
Hydrocarbons with only single bonds are called alkanes. Alkanes are called saturated hydrocarbons because each carbon is bonded with as many hydrogen atoms as possible. Potassium permanganate will not react with alkanes since they are saturated.
What colour does bromine water turn with an alkane?
An alkane undergoes no reaction with bromine water and therefore there is no colour change.
What happens when bromine water is added to hexane?
This is a substitution reaction, where one bromine atom replaces a hydrogen atom in the hexane, forming bromohexane (C6H13Br) and hydrogen bromide (HBr). The ultraviolet light powers this reaction by splitting the bromine molecules to form bromine free radicals that react with the hexane.
Is bromine soluble in water?
Bromine is a naturally occurring element that is a liquid at room temperature. It has a brownish-red color with a bleach-like odor, and it dissolves in water.
Which compound does not react with bromine water?
Cyclo-propane
Was this answer helpful?
Does bromine react with acids?
Detailed examination of the reaction of bromate with hydrochloric acid proves that, in the presence of excess of hydrochloric acid, these compounds quickly react forming bromine mono- chloride and elementary chlorine. The proportions of the reaction products have been determined.
Why are alkenes more reactive than alkanes?
Alkenes are unsaturated, meaning they contain a double bond . This bond is why the alkenes are more reactive than the alkanes .
Which type of hydrocarbons will not react with bromine quizlet?
Alkanes do not have a reaction with bromine, the red color will persist when added. Alkenes react with cold concentrated sulfuric acid by addition.
How do alkanes and alkenes react with potassium permanganate?
Alkanes and aromatic compounds do not react with potassium permanganate. and and and etc. Hydrocarbons are incompatible with water and should never be poured down the sink.
How does hydrocarbons react with water?
Hydrocarbon is converted to hydrogen and carbon monoxide through the water- carbon reaction, as shown in eq 1. Carbon monoxide, CO, is then converted to hydrogen and carbon dioxide by the water-gas shift reaction, as shown in eq 2.
Is bromine water polar or nonpolar?
The water from the bromine water is polar and therefore the non-polar organic reactant and product will not dissolve in the water; because of this, two layers form as this polar and non-polar layer do not mix.
How the reaction with bromine can be used to test for an alkene?
It is possible to use a basic bromine water test to know the difference between an alkane and an alkene. If the bromine reacts with the carbon-carbon double bond, the alkene can turn brown bromine water colourless. With alkenes and alkynes, bromine reacts quickly.
How does bromine water Decolourise with an alkene?
An alkene decolourise bromine water because the bromine reacts with the carbon-carbon double bonds. The carbon-carbon bond is broken and bromine gets attached to the alkene thus forming alkane. This is also the reason why alkene is known as unstaurated hydrocarbons.
Which of the following reaction does not give alkane?
Dehydrohalogenation of alkyl halide gives alkenes but not alkane.
How do you tell if a compound is an alkane?
The distinguishing feature of an alkane, making it distinct from other compounds that also exclusively contain carbon and hydrogen, is its lack of unsaturation. That is to say, it contains no double or triple bonds, which are highly reactive in organic chemistry.
Is alkane and alkyl same?
An alkane is a fully saturated hydrocarbon, typically with no other major substituents or heteroatoms. An alkyl group indicates a functional group that is generally only composed of carbon and hydrogen. This group is part of an organic molecule that typically has multiple functionalities.



