How do monomers make polymers?

James Craig
Published Jan 12, 2026
The monomers combine with each other using covalent bonds to form larger molecules known as polymers. In doing so, monomers release water molecules as byproducts. This type of reaction is known as dehydration synthesis, which means “to put together while losing water.”
How do monomers form a polymer?
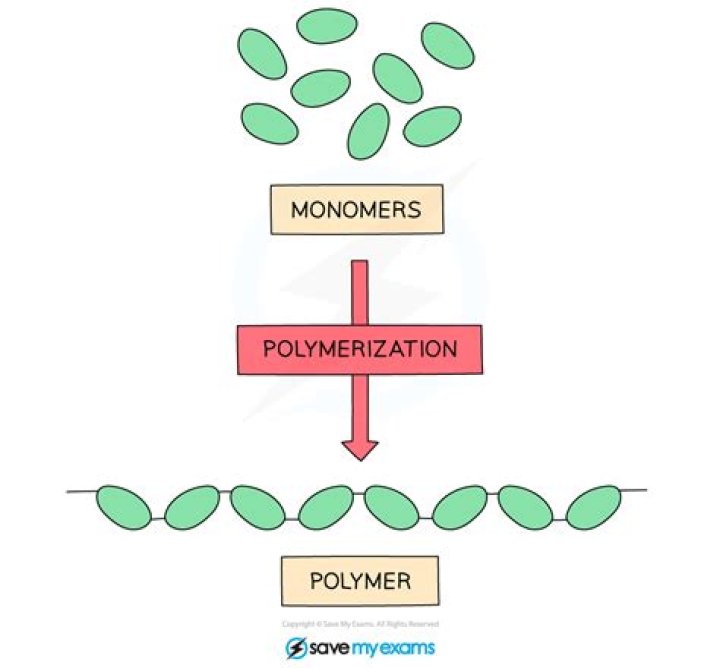
Monomers form polymers by forming chemical bonds or binding supramolecularly through a process called polymerization. Sometimes polymers are made from bound groups of monomer subunits (up to a few dozen monomers) called oligomers.
How is polymers formed?
Polymers are formed by two main ways called addition and condensation polymerization. In addition, polymerization, an initiator (or catalyst) reacts with a starting monomer. The result of this initiation reaction is a monomer attached to the initiator with an unsatisfied bond.
Do monomers make up polymers?
Polymers are a class of synthetic substances composed of multiples of simpler units called monomers. Polymers are chains with an unspecified number of monomeric units. Homopolymers are polymers made by joining together monomers of the same chemical composition or structure.
What are lipids monomers and polymers?
Monomers are smaller molecules, and when bonded together, make up polymers . -Fatty acids are the monomers for lipids , for example, and regardless of how they are bonded (as a saturated or unsaturated fat, for example), they will form lipids.
45 related questions foundWhat is in between monomers and polymers?
The examples of the relationship between monomers and polymers: Polysaccharides are polymers and their monomers are called monosaccharides. Complex carbohydrates are polysaccharides.
How are monomers made?
Monomers are small molecules that can be joined to form more complex molecules called polymers in a repeated fashion. Monomers form polymers by the formation of chemical bonds or the supramolecular binding through a process called polymerization.
How does polymers formed describe each structure?
A single polymer molecule may consist of hundreds to a million monomers and may have a linear, branched, or network structure. Covalent bonds hold the atoms in the polymer molecules together and secondary bonds then hold groups of polymer chains together to form the polymeric material.
What are polymers ks3?
Polymers are made by chemical reactions that join lots of small molecules together to make long molecules. For example, a molecule of poly(ethene) is made by joining thousands of ethene molecules together. Long molecules like these give polymers their properties.
What are polymers ks2?
A polymer is a molecule, made from joining together many small molecules called monomers. The word "polymer" can be broken down into "poly" (meaning "many" in Greek) and "mer" (meaning "unit").
What is a monomer ks3?
Polymers are very large molecules made when many smaller molecules join together, end to end. The smaller molecules are called monomers. In general: lots of monomer molecules → a polymer molecule. The polymers formed are called addition polymers.
How are polymers formed GCSE?
Polymers have very large molecules. They are formed when many small molecules join together. This process is called polymerisation. When alkenes join together to form a polymer with no other substance being produced in the reaction, the process is called addition polymerisation.
Which substances are made up of polymers?
Polymers make up many of the materials in living organisms, including, for example, proteins, cellulose, and nucleic acids. Moreover, they constitute the basis of such minerals as diamond, quartz, and feldspar and such man-made materials as concrete, glass, paper, plastics, and rubbers.
How are macromolecule polymers assembled from monomers?
Most macromolecules are made from single subunits, or building blocks, called monomers. The monomers combine with each other using covalent bonds to form larger molecules known as polymers. In doing so, monomers release water molecules as byproducts.
Why are monomers important in the formation of polymers?
monomer, a molecule of any of a class of compounds, mostly organic, that can react with other molecules to form very large molecules, or polymers. The essential feature of a monomer is polyfunctionality, the capacity to form chemical bonds to at least two other monomer molecules.
Why are monomers and polymers different?
A polymer is a macroscopic material built from a large number of repeating single units bound together. A monomer is a single repeating unit that is covalently bound to form polymers.
How polymers are different from monomers explain with examples?
Polymers are macromolecules with very high molecular weights built from many repetitive units called monomers. Monomers are simple molecules with low molecular weights. Polymers are complex molecules with very high molecular weight. A monomer can have different combination units.
How are monomers linked together?
The monomers combine with each other using covalent bonds to form larger molecules known as polymers. In doing so, monomers release water molecules as byproducts.
Are lipids polymers?
Lipids are not usually polymers and are smaller than the other three, so they are not considered macromolecules by some sources 1,2start superscript, 1, comma, 2, end superscript.
What are the 3 main types of polymers?
There are three types of classification under this category, namely, Natural, Synthetic, and Semi-synthetic Polymers.
- Natural Polymers: ...
- Semi-synthetic Polymers: ...
- Synthetic Polymers: ...
- Linear Polymers. ...
- Branched-chain Polymers. ...
- Cross-linked Polymers. ...
- Classification Based on Polymerization. ...
- Classification Based on Monomers.
What type of materials are formed when monomers join together?
polymerization, any process in which relatively small molecules, called monomers, combine chemically to produce a very large chainlike or network molecule, called a polymer.
How are condensation polymers made?
Some polymers are made via condensation polymerisation . In condensation polymerisation, a small molecule is formed as a by-product each time a bond is formed between two monomers . This small molecule is often water. An example of a condensation polymer is nylon.
What is a monomer GCSE?
A monomer is a small reactive molecule that can be joined with other monomers to form long chains. These long chain molecules can be made up from tens of thousands of monomers joined together.