Is aniline a nucleophile?

Emily Sparks
Published Jan 23, 2026
With respect to benzene, both aniline and phenol are activated nucleophiles, and often do not require Lewis acid catalysis. The lone pairs on phenol donate electron density to the entire ring, and the phenyl ring becomes MORE nucleophilic.
Is aniline an electrophile?
Aniline refers to the formation of Acetanilide by nucleophilic replacement reaction with acetic anhydride and the reaction is called acetylation. Aniline serves as the nuclepohile in this reaction and acetic anhydride group acyl (CH3CO-) acts as the electrophile.
Is aniline strong nucleophile?
Knowing what the reaction is, we can then determine that the amine species are the nucleophiles in those reactions. For aniline, resonance of the N lone pair with the adjacent π system decreases the availability if the lone pair and so makes aniline a poorer nucleophile thus resulting in the lower reactivity.
Why is aniline a good nucleophile?
Anilinium chlroide is a good nucleophile because is has a lone pair of electrons to donate. The lone paid of electrons of the nitrogen in the aniline is not entirely free since it is in resonance with the electrons f the benzene ring.
Are amines nucleophiles or electrophiles?
A nucleophile is something which is attracted to, and then attacks, a positive or slightly positive part of another molecule or ion. All amines contain an active lone pair of electrons on the very electronegative nitrogen atom.
34 related questions foundWhich of the following is not nucleophile?
BF3 is electron deficient compound. It does not have lone pair of electrons to donate. So it is not nucleophilic.
Is nh3 a nucleophile?
Ammonia doesn't carry a negative charge. But it has a lone pair of electrons. And nitrogen is more electronegative than hydrogen, so the nitrogen atom has a δ⁻ charge. So NH₃ can act as a nucleophile and attack the δ⁺ C atom of an alkyl halide.
Is phenol A nucleophile?
The phenoxide anion is a strong nucleophile with a nucleophilicity comparable to the one of carbanions or tertiary amines. It can react at both its oxygen or carbon sites as an ambident nucleophile (see HSAB theory).
Is aniline a good nucleophile is Anilinium chloride a good nucleophile explain the differences between the two?
Aniline is not a good nucleophile because the lone pair of electrons of nitrogen are resonance stabilized by the benzene ring. Anilinium chloride is also not a good nucleophile, and actually acts as an electrophile. So rather than donating electrons, it accepts.
Is bromine a good nucleophile?
As you noted bromine has a negative charge (and also lower electronegativity than oxygen) and is therefore the better nucleophile (even under these aqueous conditions).
Can NH2 be a nucleophile?
NH2(-) is a better nucleophile than NH3. HS(-) is a better nucleophile than H2S. The greater the negative charge, the more likely an atom will give up its pair of electrons to form a bond.
Are ethers nucleophilic?
Due to the high chemical stability of ethers, the cleavage of the C-O bond is uncommon in the absence of specialized reagents or under extreme conditions. In organic chemistry, ether cleavage is an acid catalyzed nucleophilic substitution reaction.
Are amides nucleophilic?
Because of their low reactivity, amides do not participate in nearly as many nucleophilic substitution reactions as other acyl derivatives do. Amides are stable to water, and are roughly 100 times more stable towards hydrolysis than esters.
Is aniline an aromatic compound?
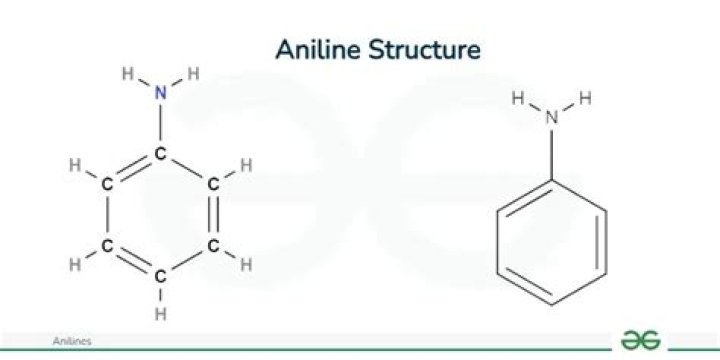
Aniline is an organic compound with the formula C6H5NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine. It is an industrially significant commodity chemical, as well as a versatile starting material for fine chemical synthesis.
Is aniline a hydrocarbon?
Aniline is the simplest aromatic amine, C6H5-NH2 and is used in the aniline point test. Aromatic hydrocarbons in oils used to prepare oil-base mud can damage elastomers and increase the toxicity of the fluid. The aniline point test is used for screening oils to infer aromatic content.
Is CN good nucleophile?
CN− is a strong nucleophile. We expect it to take part in SN2 reactions. It is also a weak base, so we do not expect either E2 or E1 eliminations. If an SN2 reaction is not possible, an SN1 reaction might occur.
Which compound is most nucleophilic?
Answer:Ethoxide is the better nucleophile. The steric bulk of t-butoxide decreases itseffectiveness as a nulcoephile. Answer : Ethoxide is the better nucleophile .
Which one of the following is the strongest nucleophile?
Hence, strongest nucleophile is EtO−
Is carbon a nucleophile?
Carbon Atom as an Extremely Strong Nucleophilic and Electrophilic Center: Dendritic Allenes Are Powerful Organic Proton and Hydride Sponges.
Is H+ A nucleophile?
H+ is one of the only electrophiles that is guaranteed to be an electrophile. It has no electrons, so of course, it can only accept electrons. Hence, it must be a lewis acid, or electrophile. OH− is almost always going to be a nucleophile, as it is negatively charged.
Is hydroxyl a nucleophile?
Hydroxyl groups in R–OH are poor nucleophiles because they're neutral and the electron pair is held tightly to the oxygen.
Is CH3 a nucleophile?
CH3- is more nucleophilic. Carbon is a smaller atom which means it will not hold a negative charge as well meaning CH3- is more reactive (more nucleophilic) than NH2-.
Is fecl3 a nucleophile?
Ferric chloride acts both as Lewis acid as well as chloride nucleophile.
Is alcl3 a nucleophile?
No it is not a nucleophile. Its is a lewis acid and extracts chlorine anion.