Is AstraZeneca COVID-19 vaccine recommended for children?

William Rodriguez
Published Jan 08, 2026
Is AstraZeneca COVID-19 vaccine recommended for children?
COVID-19 Vaccine AstraZeneca is not recommended for children aged below 18 years. No data are currently available on the use of COVID-19 Vaccine AstraZeneca in children and adolescents younger than 18 years of age.
How old do you have to be to get the AstraZeneca vaccine?
The vaccine is not recommended for persons younger than 18 years of age pending the results of further studies.
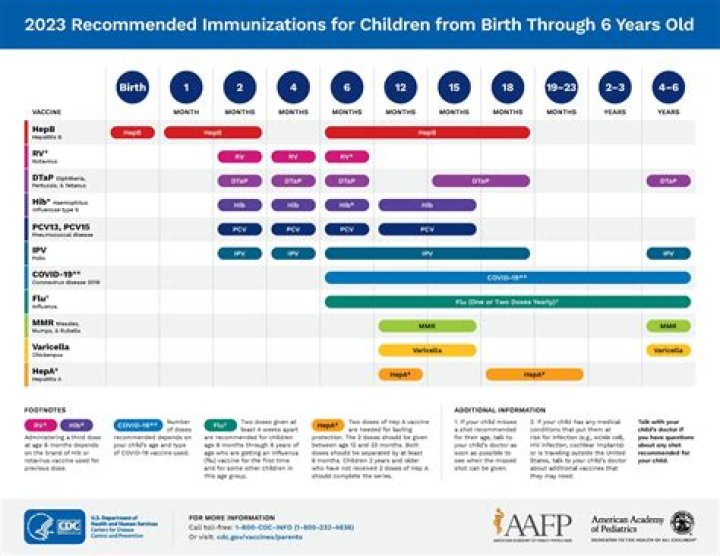
At what age can children get the COVID-19 vaccine?
The NHS is offering coronavirus (COVID-19) vaccines to children aged 5 to 11 years. Experts have advised that parents of all children aged 5 to 11 years should be offered the chance to have their child vaccinated.
What are the common side effects of the Astra Zeneca COVID-19 vaccine?
The most frequently reported adverse reactions are injection site tenderness (68%), injection site pain (58%), headache (53%), fatigue (53%), myalgia (44%), malaise (44%), pyrexia (includes feverishness (33%) and fever ≥38°C (8%)), chills (32%), arthralgia (27%) and nausea (22%).
The majority of these adverse reactions were mild to moderate in severity and usually resolved within a few days of vaccination. Very rare cases of thrombosis with thrombocytopenia syndrome have been reported post-marketing within the first three weeks following vaccination (see section 4.4).
What are some common side effects of the COVID-19 vaccine in children?
Common side effects their arm feeling heavy or sore where they had the injection feeling achy or like they have the flu feeling tired having a headache
45 related questions foundWhat are the most common adverse reactions to the covid-19 vaccine?
The most frequent adverse reactions in trials were pain at the injection site, fatigue, headache, myalgia (muscle pains), chills, arthralgia (joint pains), and fever; these were each reported in more than 1 in 10 people.
What are the common side effects of the COVID-19 vaccine?
Common side effects
- having a painful, heavy feeling and tenderness in the arm where you had your injection. This tends to be worst around 1 to 2 days after the vaccination
- feeling tired
- headache
- general aches, or mild flu like symptoms
How does the AstraZeneca COVID-19 vaccine work?
COVID-19 Vaccine AstraZeneca stimulates the body's natural defences (immune system). It causes the body to produce its own protection (antibodies) against the virus. This will help to protect you against COVID-19 in the future. None of the ingredients in this vaccine can cause COVID-19.
Can you get a fever or temperature after the COVID-19 vaccine?
Although a fever can occur within a day or two of vaccination, if you have any other COVID-19 symptoms or your fever lasts longer, stay at home and arrange to have a test.
What is the recommended dosing interval for the AstraZeneca COVID-19 vaccine?
AstraZeneca COVID-19 vaccine dosing interval. The recommended interval between 2 doses of the AstraZeneca COVID-19 vaccine is 12 weeks. The minimum interval between doses is 4 weeks. The duration of protection after a single dose of AstraZeneca has not yet been established.
Is the Pfizer COVID-19 vaccine available for children at high risk?
About the vaccine. Children will be offered the Pfizer COVID-19 vaccine. Each vaccine is a third of the dose of vaccine that is given to older children and adults. Your child needs 2 injections of the vaccine usually 8 weeks apart. The vaccine has been tested to make sure it is as safe as possible.
Do children need parental consent for the COVID-19 vaccine?
All parents or those with parental responsibility are asked for consent and will usually make this decision, jointly with their children. The information leaflet is addressed to the child (as the recipient of the vaccine) and encourages them to discuss the decision about the vaccine with their parents.
In secondary schools, some older children may be sufficiently mature to provide their own consent. This sometimes occurs if a parent has not returned a consent form but the child still wishes to have the vaccine on the day of the session. Every effort will be made to contact the parent to seek their verbal consent. The school has no role in this process.
Is there a COVID-19 vaccination for young people of 16 and 17 years of age in UK?
Health and Social Care Secretary Sajid Javid accepts advice from JCVI to offer COVID-19 vaccination to those aged 16 and 17.
Who can get a spring booster for the coronavirus disease?
People aged 75 years and older, residents in care homes for older people, and those with weakened immune systems will be offered a spring booster of coronavirus (COVID-19) vaccine. Appointments will be available from the National Booking Service shortly.
Is the AstraZeneca COVID-19 vaccine available in the UK?
To date, supply of the AstraZeneca vaccine in the UK has been authorised on a temporary basis by the MHRA under Regulation 174 of the Human Medicine Regulations 2012, but as this was always intended to be a temporary arrangement, supply of this vaccine will change to be in accordance with the CMAs.
Does the COVID-19 AstraZeneca vaccine contain any ingredients that can cause COVID-19?
COVID-19 Vaccine AstraZeneca stimulates the body's natural defences (immune system). It causes the body to produce its own protection (antibodies) against the virus. This will help to protect you against COVID-19 in the future. None of the ingredients in this vaccine can cause COVID-19.
Can you still get Covid after vaccine?
Like all medicines, no vaccine is completely effective, so you should continue to take recommended precautions to avoid infection. Some people may still get COVID-19 despite having a vaccination, but this should be less severe.
What are the possible side effects of the BNT162b2 COVID-19 vaccine?
The most common side effects with BNT162b2 (which may affect more than 1 in 10 people) were pain at the injection site, tiredness, headache, muscle pain, chills, joint pain and fever.
Is temperature check a reliable way to detect coronavirus disease?
There is little scientific evidence to support temperature screening as a reliable method for detection of COVID-19 or other febrile illness, especially if used as the main method of testing.
Temperature readings from temperature screening systems will measure skin temperature rather than core body temperature. In either case, natural fluctuations in temperature can occur among healthy individuals. These readings are therefore an unreliable measure for detection of COVID-19 or other diseases which may cause fever. Furthermore, infected people who do not develop a fever or who do not show any symptoms would not be detected by a temperature reading and could be more likely to unknowingly spread the virus.
Can you catch COVID-19 after 2 doses of vaccines?
Although 2 doses of vaccine will greatly reduce your own risk of becoming unwell with COVID-19, it is still possible to contract the virus and pass it to others.
How far apart should you wait between the doses of the Oxford COVID-19 vaccine?
The MHRA authorisation includes conditions that the AstraZeneca (Oxford) vaccine should be administered in 2 doses, with the second dose given between 4 and 12 weeks after the first.
What is the maximum interval for receiving the Pfizer-BioNTech and AstraZeneca COVID-19 vaccine?
The committee supports a 2-dose vaccine schedule for the Pfizer-BioNTech and AstraZeneca vaccines. Given the data available, and evidence from the use of many other vaccines, JCVI advises a maximum interval between the first and second doses of 12 weeks for both vaccines.
Is the COVID-19 vaccine safe?
The MHRA confirmed on 9 September 2021 that the COVID-19 vaccines made by Pfizer and AstraZeneca can be used as safe and effective booster doses. Following review of data for the COVID-19 Vaccine Moderna vaccine, the MHRA and CHM experts also concluded that this vaccine can be used as a safe and effective booster dose.
All vaccines and medicines have some side effects. These side effects need to be continuously balanced against the expected benefits in preventing illness. The COVID-19 Pfizer/BioNTech Vaccine was evaluated in clinical trials involving more than 44,000 participants. The most frequent adverse reactions in these trials
How long after having COVID-19 can I get the spring booster vaccine?
If you are unwell, wait until you have recovered to have your vaccine. If you have had confirmed COVID-19 you should ideally wait 4 weeks before having your spring booster.
Can the COVID-19 vaccine affect your ability to drive and use machines?
Driving and using machines
COVID-19 mRNA Vaccine BNT162b2 has no or negligible influence on the ability to drive and use machines. However, some of the effects mentioned under section 4 'Possible side effects' may temporarily affect the ability to drive or use machines.



