Is bromine water organic or inorganic?

Noah Mitchell
Published Jan 23, 2026
Bromine in the environment
Is bromine water organic?
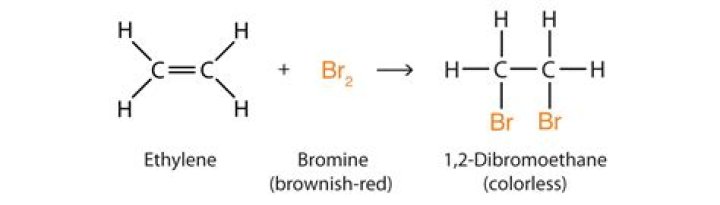
Organic compounds like phenols, alkenes, acetyl compounds and anilines will readily undergo bromine water tests. During the process, change in the colour of the bromine water indicates the presence of an unsaturated group in the organic compound.
What is the difference between bromine and bromine water?
Pure Bromine is highly reactive and when added to water it's results are explosive. However, Bromine water means bromine already diluted with water which is lesser explosive than bromine in water.
What is bromine water made of?
Bromine water is an oxidizing, intense yellow-to-red mixture containing diatomic bromine (Br2) dissolved in water (H2O).
What happens when bromine is dissolved in water?
Bromine can easily be dissolved in water (35 g per L water), carbon disulfide and other organic solutions. When added to water, bromine forms hypobromous acid. Hyprobromous acid is a weak acid. It partly dissociates to form hydrogen ions and hypobromite ions.
26 related questions foundIs bromine soluble in water?
Bromine is a naturally occurring element that is a liquid at room temperature. It has a brownish-red color with a bleach-like odor, and it dissolves in water.
Does bromine purify water?
Bromine is another halogen used to disinfect water. Though not approved for use in municipal drinking water, bromine has been recognized by the USEPA as an effective and permissible disinfectant for drinking water since 1976.
Is bromine water bromine solution?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot.
Is bromine water an oxidizing agent?
The solution of Bromine in water is known as bromine water. Like chlorine water, it is also a good oxidizing agent, and it is more useful because it does not decompose so readily. It liberates free iodine from iodide-containing solutions and sulfur from hydrogen sulfide.
What is bromine water a test for?
A simple test with bromine water can be used to tell the difference between an alkane and an alkene. An alkene will turn brown bromine water colourless as the bromine reacts with the carbon-carbon double bond. In fact this reaction will occur for unsaturated compounds containing carbon-carbon double bonds.
What is the purpose of bromine water test for lipids?
Bromine water test
Bromine water becomes colourless when shaken with an unsaturated vegetable oil, but it stays orange-brown when shaken with a saturated vegetable fat. Bromine water can also be used to determine the level of saturation of a vegetable oil.
What concentration is bromine water?
Bromine Water, Saturated, approximately 3% (w/v), Ricca Chemical | Fisher Scientific.
How do you identify bromine in an organic compound?
A sensitive test for bromine is the reaction with fluorescein to give a deep red colour caused by bromination of the organic molecule, or by its reaction with fuchsine dyes in the presence of sulfurous acid, to give a deep blue colour.
Is bromine water polar or nonpolar?
The water from the bromine water is polar and therefore the non-polar organic reactant and product will not dissolve in the water; because of this, two layers form as this polar and non-polar layer do not mix.
Is bromine aqueous or liquid?
Bromine is a dark reddish-brown fuming liquid with a pungent odor. Denser than water and soluble in water. Hence sinks in water.
How does bromine water test for alkanes?
You can use bromine water, which is an orange solution, to distinguish between alkanes and alkenes. There is no change when bromine water is mixed with an alkane, but it turns colourless when mixed with an alkene.
What color is bromine water?
Bromine water contains bromine molecules, Br2 , dissolved in water. Pure bromine has a very dark color, so bromine water should be yellowish to light orange in color.
How do you make bromine water from bromine liquid?
Add 0.5 ml of bromine to 100 ml of water. Or crush an ampoule under 200 ml of water and decant the liquid into a bottle. Refer to CLEAPSS Hazcard HC015b and Recipe Book RB017 for alternative method.
Is bromine a solvent or solute?
Explanation: Bromine is a nonpolar solvent. Only nonpolar molecules will be soluble in or miscible with bromine. Thus, only the following nonpolar molecules will be soluble in or miscible with bromine: C5H12 (l), vegetable oil, and I2 (s).
Which compound does not react with bromine water?
Cyclo-propane
Was this answer helpful?
Can I use bromine in my swimming pool?
Yes, bromine tabs can be used in outdoor pools, but the problem with bromine is that it cannot be stabilized or protected from the sun with cyanuric acid. For outdoor pools that receive strong direct sunlight, bromine levels can deplete rapidly, requiring more bromine to maintain healthy levels.
Which is safer chlorine or bromine?
Bromine: Is gentler on your skin than chlorine, but can be a bit more difficult to wash off after a long soak. If you or anyone who uses your hot tub on a regular basis has sensitive skin or any kind of upper respiratory difficulties, bromine will likely be the better choice.
What is soluble in liquid bromine?
Bromine is slightly soluble in water, and highly soluble in carbon disulfide, aliphatic alcohols (such as methanol), and acetic acid. It bonds easily with many elements and has a strong bleaching action. Bromine is highly reactive and is a powerful oxidizing agent in the presence of water.