Is hydration of alkenes electrophilic addition?

Emily Ross
Published Jan 17, 2026
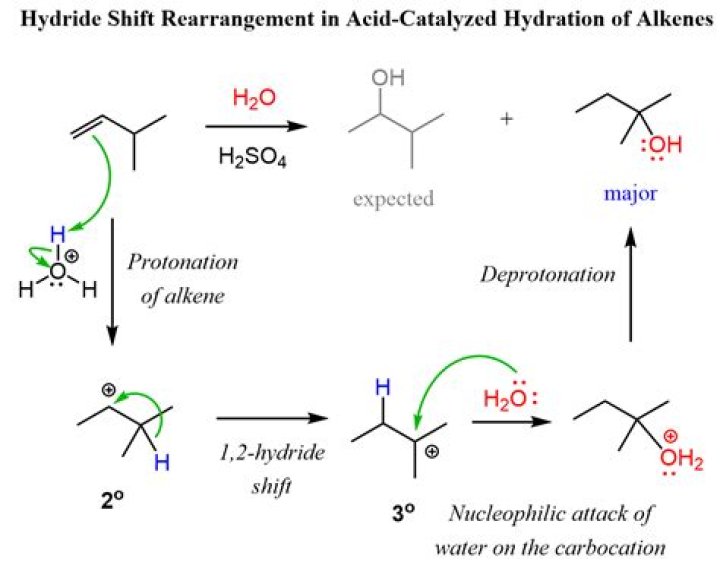
Electrophilic hydration is the act of adding electrophilic hydrogen from a non-nucleophilic strong acid (a reusable catalyst, examples of which include sulfuric and phosphoric acid) and applying appropriate temperatures to break the alkene's double bond.
Is hydration a nucleophilic addition?
Acid or Base Catalyzed Hydrate Formation
The addition of an acid or base changes the mechanism and promote the nucleophilic addition of water to a carbonyl to form a hydrate.
Is hydration nucleophilic or electrophilic?
Yes, water is a nucleophile. Water is both a nucleophile and an electrophile. and as electrophile by giving a proton to a nucleophile.
Is hydration an addition reaction?
Alkenes undergo an addition reaction with water in the presence of a catalyst to form an alcohol. This type of addition reaction is called hydration. The water is added directly to the carbon – carbon double bond.
Is hydrogenation of alkenes electrophilic addition?
Hydrogenation. There is another reaction of alkenes, hydrogenation, which deserves mention but which is not related to the electrophilic addition mechanism. Hydrogenation is the addition of molecular hydrogen (H22) to the alkene double bond. This converts a simple alkene into an alkane.
35 related questions foundIs hydration electrophilic addition?
Electrophilic hydration is the act of adding electrophilic hydrogen from a non-nucleophilic strong acid (a reusable catalyst, examples of which include sulfuric and phosphoric acid) and applying appropriate temperatures to break the alkene's double bond.
What is hydration of alkenes?
Hydration of Alkenes
The net addition of water to alkenes is known as hydration. The result involves breaking the pi bond in the alkene and an OH bond in water and the formation of a C-H bond and a C-OH bond.
What type of reaction is hydration?
A hydration reaction is a chemical reaction where a hydrogen and hydroxyl ion is attached to a carbon in a carbon double bond. Generally, one reactant (usually an alkene or alkyne) reacts with water to yield ethanol, isopropanol, or 2-butanol (all alcohols) are a product.
Can alkenes form addition polymers?
Alkenes can be used to make polymers . Polymers are very large molecules made when many smaller, reactive molecules join together, end to end.
Does hydration of alkenes require heat?
Heat is used to catalyze electrophilic hydration; because the reaction is in equilibrium with the dehydration of an alcohol, which requires higher temperatures to form an alkene, lower temperatures are required to form an alcohol. The exact temperatures used are highly variable and depend on the product being formed.
What is the product of the hydration of an alkene?
Addition of Water to Alkenes (Hydration of Alkenes)
However, with the presence of small amount of an acid, the reaction does occur with a water molecule added to the double bond of alkene, and the product is an alcohol.
Is hydration and hydrolysis the same?
The difference between hydration and hydrolysis is that hydrolysis is the process of breaking of compounds using water, whereas hydration is defined as the electrophilic addition reaction, and there is no cleavage of the original molecule.
Is hydrogenation an example of electrophilic addition?
Is hydrogenation electrophilic addition? There is another alkene reaction, hydrogenation, which merits notice but is not related to the mechanism of electrophilic addition. The addition of molecular hydrogen (H2) to the alkene double bond is termed hydrogenation. It transforms a simple alkene into an alkane.
What is meant by nucleophilic addition reaction?
In organic chemistry, a nucleophilic addition reaction is an addition reaction where a chemical compound with an electrophilic double or triple bond reacts with a nucleophile, such that the double or triple bond is broken.
Which of the following shows the highest nucleophilic addition reaction?
Hence, option 3 is the most reactive for nucleophilic addition reaction.
What happens when water is added to ketone?
Aldehydes and ketones react with water to give 1,1-geminal diols known as hydrates.
How do alkenes form addition polymers?
During the addition polymerization process, the pi bonds of the double bonds in each alkene molecule sort of break open, thereby allowing the free bonds to join with one another to form a chain known as poly(alkene) or polythene. This chain contains units that repeat themselves, known as repeating units.
What type of polymers are formed from alkenes?
This page looks at the polymerisation of alkenes to produce polymers like poly(ethene) (usually known as polythene, and sometimes as polyethylene), poly(propene) (old name: polypropylene), PVC and PTFE. It also looks briefly at how the structure of the polymers affects their properties and uses.
How are polymers formed from alkene monomer?
An addition reaction is one in which two or more molecules join together to give a single product. During the polymerisation of ethene, thousands of ethene molecules join together to make poly(ethene) - commonly called polythene. Ethene is known as the monomer. Poly(ethene) is the polymer.
Is hydration of alkenes syn or anti addition?
The hydroboration mechanism has the elements of both hydrogenation and electrophilic addition and it is a stereospecific (syn addition), meaning that the hydroboration takes place on the same face of the double bond, this leads cis stereochemistry.
What is hydration reaction in chemistry?
In chemistry, a hydration reaction is a chemical reaction in which a substance combines with water. In organic chemistry, water is added to an unsaturated substrate, which is usually an alkene or an alkyne. This type of reaction is employed industrially to produce ethanol, isopropanol, and butan-2-ol.
What is an example of a hydrate?
Other examples of hydrates are Glauber's salt (sodium sulfate decahydrate, Na2SO4∙10H2O); washing soda (sodium carbonate decahydrate, Na2CO3∙10H2O); borax (sodium tetraborate decahydrate, Na2B4O7∙10H2O); the sulfates known as vitriols (e.g., Epsom salt, MgSO4∙7H2O); and the double salts known collectively as alums (M+2 ...
Is Hydrohalogenation an electrophilic addition reaction?
A hydrohalogenation reaction is the electrophilic addition of hydrohalic acids like hydrogen chloride or hydrogen bromide to alkenes to yield the corresponding haloalkanes.
How are the following compound obtained by hydration of alkene?
1 Answer. (1) When ethene is passed through cold 98 % H2SO4 , ethyl hydrogen sulphate is formed, which on heating with water gives ethanol. (2) Propene with cold 80% H2SO4 gives isopropyl hydrogen sulphate which further on boiling with water gives isopropyl alcohol.
Is the hydration of ethene electrophilic addition?
The mechanism for the hydration of ethene
This assumes that you know about the electrophilic addition reactions of ethene, and about the use of curly arrows in organic reaction mechanisms.