What concentration is bromine water?

Rachel Ellis
Published Jan 21, 2026
Bromine Water, Saturated, approximately 3% (w/v), Ricca Chemical | Fisher Scientific.
Is bromine water a solution?
Bromine water, also called Bromide bromate solution or Bromine solution with the chemical formula Br2. The molecular weight of bromine water is 159.81 and the density is 1.307 g/mL. Bromine water is a yellow mixture solution with high oxidizing property, prepared by dissolving diatomic bromine (Br2) in water (H2O).
Is bromine water the same as bromine solution?
Bromine water is a reagent which is used to test for unsaturation in organic compound. It is 2.8 % bromine in water. In many places, it is refer to as bromine solution. But it is observed that adding bromine to water gives hypobromous acid (HOBr) and hydrobromic acid (HBr) via a disproportionation reaction.
What is the solubility of bromine in water?
Bromine is readily soluble in water (0.33 mg / ml), but is very soluble in organic solvents such as CCl4. As Ram Kowshik mentioned, due to the relatively larger size of the molecule, water can induce a dipole on Br2.
What is meant by bromine water?
Bromine water is an oxidizing, intense yellow-to-red mixture containing diatomic bromine (Br2) dissolved in water (H2O).
16 related questions foundWhy bromine is slightly soluble in water?
It's solubility can be explained by the tendency of Bromine to polarize itself. Thereby, when a polar H2O molecule interacts with Br2, the Oxygen atom will induce a positive dipole thereby resulting in dipole-induced dipole interactions. This results in Bromine's solubility in water.
Does bromine mix with water?
Bromine reacts with water to produce hypobromite, OBr-. The pH of the solution determines the position of the equilibrium. Bromine is not reactive towards oxygen or nitrogen but it will react ozone at -78°C to form the unstable compound bromine(IV) oxide.
Is bromine water organic or inorganic?
Bromine is a naturally occurring element that can be found in many inorganic substances. Humans however, have many years ago started the introduction of organic bromines in the environment. These are all compounds that are not natural and can cause serious harm to human health and the environment.
Can bromine water oxidizes alcohol?
1 Answer. Since Alcohol can not be oxidized so Alcohol can not decolourise Bromine water. Aldehyde decolourises Bromine water.
Do ketones give bromine water test?
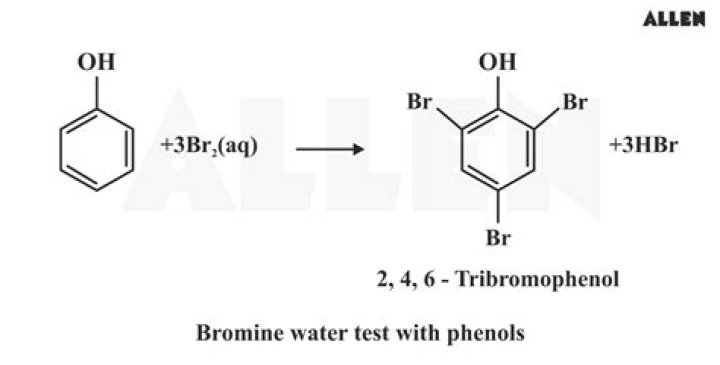
Bromine water (Br2/H2O) test is used to distinguish aldehydes (RCHO) from ketones (RCOR') especially carbohydrates Aldoses (Glucose) from Ketoses (Fructose). Bromine water chemo selectively oxidizes aldehydes (RCHO) into Acids (RCOOH) while ketones and alcohols (ROH) groups are unreactive.
How do you make bromine water from bromine liquid?
Add 0.5 ml of bromine to 100 ml of water. Or crush an ampoule under 200 ml of water and decant the liquid into a bottle. Refer to CLEAPSS Hazcard HC015b and Recipe Book RB017 for alternative method.
What is bromine water a test for?
A simple test with bromine water can be used to tell the difference between an alkane and an alkene. An alkene will turn brown bromine water colourless as the bromine reacts with the carbon-carbon double bond. In fact this reaction will occur for unsaturated compounds containing carbon-carbon double bonds.
Is bromine water an oxidizing agent?
The solution of Bromine in water is known as bromine water. Like chlorine water, it is also a good oxidizing agent, and it is more useful because it does not decompose so readily. It liberates free iodine from iodide-containing solutions and sulfur from hydrogen sulfide.
Why is bromine added to water?
Bromine substances are disinfectants and can be used as an alternative for chlorine. In swimming pools, bromine is used against the formation and growth of algae, bacteria and odors in swimming water. In the United States, bromine has been used since 1936 to treat swimming water.
What is the color of bromine water?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot.
What is soluble in liquid bromine?
Bromine is slightly soluble in water, and highly soluble in carbon disulfide, aliphatic alcohols (such as methanol), and acetic acid. It bonds easily with many elements and has a strong bleaching action. Bromine is highly reactive and is a powerful oxidizing agent in the presence of water.
Is bromine denser than water?
Bromine is a dark reddish-brown fuming liquid with a pungent odor. Denser than water and soluble in water.
What is the density of bromine G cm3?
Density of Bromine is 3.12g/cm3. Typical densities of various substances at atmospheric pressure.
How do you make bromine water from potassium bromide?
Bromine Solution Preparation
Dissolve 3 g of potassium bromate and 15 g of potassium bromide in sufficient water to produce 1000 ml.
Which of the following will give bromine water test?
Ethene, butyne, and hexyne can decolourize bromine water. The bromine loses its original red-brown color to give a colorless liquid. The decoloration of bromine is often used as a test for C-C double and triple bond.