What is aldehyde formula?

Mia Kelly
Published Jan 16, 2026
The chemical formula for an aldehyde is RCHO. In this formula, R represents a hydrogen atom or carbon/hydrogen chain, CO represents the carbonyl, and H represents the hydrogen attached to the carbonyl chain.
How do you write aldehyde formula?
In aldehydes, the carbonyl group has one hydrogen atom attached to it together with either a 2nd hydrogen atom or a hydrogen group which may be an alkyl group or one containing a benzene ring. The general formula of alkene is CnH2n+1 so the general formula for aldehyde will be CnH2n+1CHO or CnH2nO.
What is aldehyde?
Aldehydes are present in many organic materials, everything from rose, citronella, vanilla and orange rind. Scientists also can create these compounds synthetically to use as ingredients for sweet-smelling perfumes and colognes.
What is the general formula for aldehyde and ketones?
CnH2n+4O.
What is Ketone formula?
Ketones contain a carbonyl group (a carbon-oxygen double bond). The simplest ketone is acetone (R = R' = methyl), with the formula CH3C(O)CH3. Many ketones are of great importance in biology and in industry. Examples include many sugars (ketoses), many steroids (e.g., testosterone), and the solvent acetone.
23 related questions foundWhat is aldehyde and ketone?
Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O. The carbon atom of this group has two remaining bonds that may be occupied by hydrogen or alkyl or aryl substituents.
What is suffix for aldehyde?
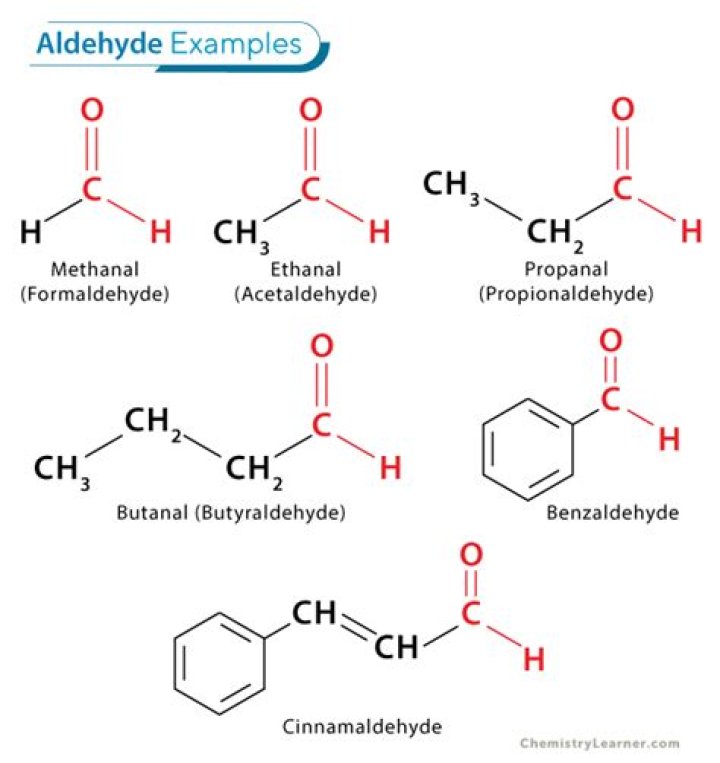
The IUPAC system of nomenclature assigns a characteristic suffix -al to aldehydes. For example, H2C=O is methanal, more commonly called formaldehyde.
What is the common name of aldehyde?
The common names of aldehydes are taken from the names of the corresponding carboxylic acids: formaldehyde, acetaldehyde, and so on. The common names of ketones, like those of ethers, consist of the names of the groups attached to the carbonyl group, followed by the word ketone.
What are aldehydes used in?
It is used in tanning, preserving, and embalming and as a germicide, fungicide, and insecticide for plants and vegetables, but its largest application is in the production of certain polymeric materials.
What are aldehydes in perfumery?
Aldehydes are the result of partial oxidation and the moniker given to them is often derived from the name of the acid it forms. They are used in the manufacture of synthetic resins and for the creation of dyestuffs, flavorings, perfumes, and other chemicals. Some are utilized as preservatives and disinfectants.
What is the condensed formula of aldehyde?
The generic condensed formula for aldehydes is RCHO (CHO is our aldehyde CHUM) and RCOR' for ketones (no cute memorization aid - if you have one please share it.)
How do you write the Iupac name of aldehyde?
Naming Aldehydes
According to the IUPAC system of nomenclature -al is attached as a suffix to parent alkane for the naming of aldehydes. For example, H2C=O is named as per the IUPAC system as methanal, commonly known as formaldehyde.
What are aldehydes 12?
Aldehydes are the organic compounds in which carbonyl group is attached to one hydrogen atom and one alkyl or aryl group.
What is aldehyde give example?
Aldehydes are given the same name but with the suffix -ic acid replaced by -aldehyde. Two examples are formaldehyde and benzaldehyde. As another example, the common name of CH2=CHCHO, for which the IUPAC name is 2-propenal, is acrolein, a name derived from that of acrylic acid, the parent carboxylic acid.
What is the homologous series of aldehyde?
A homologous series for aldehydes is methanal, ethanal, propanal, and butanal with chemical formulas of HCHO, CH3CHO, CH3CH2CHO, and CH3CH2CH2CHO, respectively where each successive compound differs from the previous one by a –CH2group.
Is cyclohexanone a ketone or aldehyde?
Cyclohexanone is a cyclic ketone that consists of cyclohexane bearing a single oxo substituent.
What is secondary suffix for aldehyde?
For aldehyde, the secondary suffix for an aldehyde is -al.
How many functional groups are there?
What are the four functional groups? In biological molecules, some of the essential functional groups include hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl groups. These groups play a significant role in forming molecules such as DNA, proteins, carbohydrates, and lipids.
What is meant by tollens reagent?
Tollens' reagent (chemical formula. ) is a chemical reagent used to distinguish between aldehydes and ketones along with some alpha-hydroxy ketones which can tautomerize into aldehydes.
What is the difference between aldehyde and alcohol?
Aldehydes and alcohols are organic compounds. They have different functional groups, resulting in different chemical and physical properties. The key difference between aldehyde and alcohol is that aldehyde contains –CHO functional group, whereas alcohol contains –OH functional group.
Is tollens a reagent?
Tollens' reagent is a chemical reagent used to determine the presence of an aldehyde, aromatic aldehyde and alpha-hydroxy ketone functional groups. The reagent consists of a solution of silver nitrate and ammonia.
What is ketone used for?
Many complex organic compounds are synthesized using ketones as building blocks. They are most widely used as solvents, especially in industries manufacturing explosives, lacquers, paints, and textiles. Ketones are also used in tanning, as preservatives, and in hydraulic fluids.