What is alkane formula?

Daniel Moore
Published Jan 24, 2026
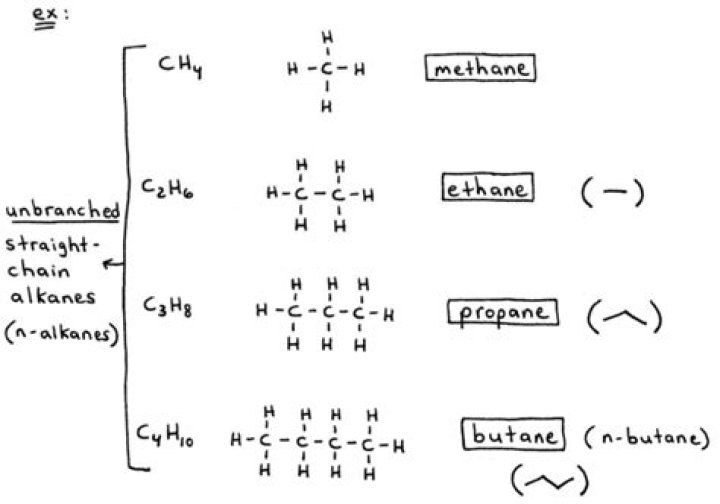
The general formula for the alkanes is C nH 2n +2, where n is the number of carbon atoms in the molecule.
What is a alkane?
Alkanes are saturated hydrocarbons. This means that their carbon atoms are joined to each other by single bonds. This makes them relatively unreactive, apart from their reaction with oxygen in the air - which we call burning or combustion. Butane. Like other homologous series, the alkanes show isomerism.
What are alkanes examples?
Alkanes are saturated hydrocarbons. By saturated hydrocarbons, it means alkanes have single hydrogen and carbon atoms in their chemical formula. Formula of alkane is CnH2n+2. Methane, propane, ethane, and butane are four alkanes.
What is alkyne formula?
Alkynes are hydrocarbons which contain carbon-carbon triple bonds. Their general formula is CnH2n-2 for molecules with one triple bond (and no rings).
What is the formula for alkanes and alkenes?
Alkanes have the general formula of CnH2n+2 where n is the number of carbon atoms. Alkenes have the general formula CnH2n.
34 related questions foundWhat is alkane alkyne and alkene?
Hydrocarbons in which one or more of the carbon atoms in the chain are bonded to another by a double or a triple bond are said to be unsaturated. The two families of unsaturated hydrocarbons are alkenes and alkynes. Alkenes of hydrocarbons has one double bond between carbon atoms.
Is c6h12 alkane alkene or alkyne?
Is C6H12 an alkane or alkene ? It has all single bonds so it must be alkane but it does not fits to the Generel Formulae of alkanes(CnH2n-1) and instead fits to the General Formulae of alkenes(CnH2n).
Where are alkynes found?
Alkynes occur in some pharmaceuticals, including the contraceptive noretynodrel. A carbon–carbon triple bond is also present in marketed drugs such as the antiretroviral Efavirenz and the antifungal Terbinafine. Molecules called ene-diynes feature a ring containing an alkene ("ene") between two alkyne groups ("diyne").
What is example of alkyne?
Ethyne aka Acetylene
Ethyne is the simplest of the alkynes; its molecular formula is C2H2, and it consists of two carbon atoms triple-bonded to each other with a hydrogen atom bonded to each of the carbons. At room temperature it's a colorless gas.
What does alkyl stand for?
Definition of alkyl
(Entry 1 of 2) : having a monovalent organic group and especially one CnH2n+1 (such as methyl) derived from an alkane (such as methane)
What is the another name of alkane?
The trivial (non-systematic) name for alkanes is 'paraffins'. Together, alkanes are known as the 'paraffin series'.
What are the 4 types of hydrocarbons?
Hydrocarbon molecules have one or more central carbon atoms in a branched or chain-like structure, surrounded by hydrogen atoms. There are four main categories of hydrocarbons: Alkanes, Alkenes, Alkynes, and Aromatic hydrocarbons.
What is the general formula for Cycloalkene?
Answer ⤵ :- Cycloalkynes have the general formula C2H2n - 4 . :- Cycloalkenes have the general formula CnH2(n-m) where m represents the number of double bonds in structure of compound .
What is an alkane ks4?
Alkanes are saturated hydrocarbons. This means that their carbon atoms are joined to each other by single bonds.
How are alkanes made?
Step 1: Position the three carbon atoms in a row. Step 2: Join the carbon atoms by single bonds. Step 3: Place four single bonds around each carbon atom. Step 4: Attach a hydrogen atom to each 'empty' bond of a carbon atom.
What is the name of simplest alkane?
Methane is the simplest alkane molecule and it is composed of one carbon and four hydrogen atoms bonded covalently. If we replace one hydrogen atom with another atom or molecule, we will form a compound commonly known as the methyl compound.
What are types of alkyne?
An alkyne is one of the four main types of hydrocarbons. Alkynes must contain at least one carbon to carbon triple bond in their chain.
...
The three simplest alkynes are ethyne (C2H2), propyne (C3H4) and butyne (C4H6).
- Ball and Stick Model of the Three Simplest Alkynes.
- Figure 1. Ethyne.
- Figure 2. Propyne.
- Figure 3. Butyne.
What is the first alkyne?
They are unsaturated hydrocarbons. The first member of alkyne family is CH≡CH; Ethyne.
Why is alkynes called acetylene?
Why alkyne is called acetylene? With regard to hydrogen atoms, because the compound is unsaturated, the extra electrons are exchanged by 2 carbon atoms that form double bonds. Alkynes from the first compound in the sequence are also commonly known as ACETYLENES.
What is the common name of alkyne?
Here, the IUPAC name is five-methyl-two-hexyn-one-ol. For common names, alkynes adopt the parent name like acetylene, with the substituents' names added as a prefix. Thus, two-butyne becomes dimethylacetylene, and two-five-dimethyl-three-hexyne would be diisopropyl acetylene.
Where are alkenes used?
What are alkenes used for? In manufacturing, alkenes find many different applications. They are used in the synthesis of alcohols, plastics, lacquers, detergents, and fuels as starting materials. For the chemical industry, the most important alkenes are ethene, propene, and 1,3-butadiene.
What is the another name of alkene?
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds. Two general types of monoalkenes are distinguished: terminal and internal. Also called α-olefins, terminal alkenes are more useful.
What does hexene smell like?
1-Hexene is a colorless liquid with a gasoline-like odor. It is used in fuels and to make flavors, perfumes, dyes, and plastic resins.
What are alkenes and alkynes?
An alkene is a hydrocarbon with one or more carbon-carbon double covalent bonds. An alkyne is a hydrocarbon with one or more carbon-carbon triple covalent bonds.