What is alkene class 11th?

Emily Ross
Published Jan 14, 2026
Acyclic unsaturated hydrocarbons containing a carbon-carbon double bond are called alkenes. They are also called olefins since the lower members of this class produce oily products on reaction with halogens such as chlorine and bromine.
What do you mean by alkene?
In chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond. Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.
What are alkenes with example?
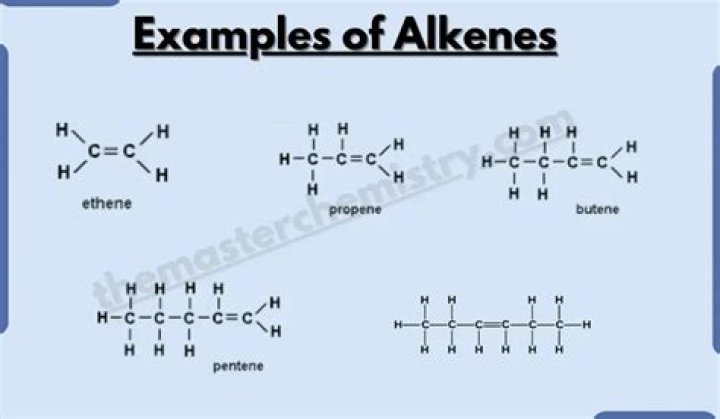
Alkenes are a class of hydrocarbons (e.g, containing only carbon and hydrogen) unsaturated compounds with at least one carbon-to-carbon double bond. Another term used to describe alkenes is olefins. Alkenes are more reactive than alkanes due to the presence of the double bond.
What is alkene formula?
The general formula for the alkenes is C nH 2n, where n is the number of carbon atoms in the molecule. Worked example. Decene is an alkene. Its molecules contain 10 carbon atoms .
Why alkene is called?
Since the compound is unsaturated with respect to hydrogen atoms, the extra electrons are shared between 2 Carbon atoms forming double bonds in alkenes. Alkenes are also called Olefins because they form oily liquids on reaction with Chlorine gas.
37 related questions foundWhat is alkene group?
Hydrocarbons that contain one or more carbon-carbon double bonds are called alkenes. They are named the same way as alkanes except that an -ene ending is used and the location of the double bond is identified.
What is alkene functional group?
The functional group in an alkene is a carbon-carbon double bond. The functional group in an alkyne is a carbon-carbon triple bond. Aromatics are cyclic strcutures that are planar, fully conjugated and that possess an odd number of electron pairs in the π bonding system.
Where is alkene found?
Alkenes are the raw materials for a number of plastics such as polyethylene, PVC, polypropylene, and polystyrene. Alkene chemistry is found in unsaturated fats, beta-carotene, and seeing light through vision. Boiling points depend on chain length, slightly less than alkanes.
Is alkene saturated or unsaturated?
The alkenes are unsaturated. This means that they have a carbon to carbon double bond. The alkanes are saturated because they only contain single bonds.
What is alkyne Class 11?
Alkynes or Acetylene
Acyclic unsaturated hydrocarbons containing a carbon-carbon triple bond are called alkynes or acetylenes. Their general formula is CnH2n-2 where n=2,3,4…. etc.
Why is an alkene unsaturated?
1 Answer. Alkenes and alkynes are called unsaturated compounds because the carbon atoms that they contain are bonded to fewer hydrogen atoms than they can possibly hold.
What is the old name of alkenes?
The old name of alkenes is Olefins.
Are alkenes polar?
Alkenes are nonpolar, since they contain nothing but carbon-carbon and carbon-hydrogen bonds, and are not soluble in water; they are also generally less dense than water.
What is alkene homologous series?
Alkene Homologous Series
The alkenes form a homologous series. A hydrocarbon homologous series is a series of hydrocarbons which: Have the same general formula. Differ by CH2 in molecular formulae from neighbouring compounds. Show a gradual variation in physical properties i.e. boiling and melting point.
Are alkenes reactive?
Alkenes are relatively stable compounds, but are more reactive than alkanes because of the reactivity of the carbon–carbon π-bond. Most reactions of alkenes involve additions to this π bond, forming new single bonds.
Do alkenes burn in air?
Alkenes combust, but they are less likely than alkanes to combust completely. Complete combustion of alkenes produces carbon dioxide and water, provided there is a plentiful supply of oxygen. Incomplete combustion of alkenes occurs where oxygen is limited and produces water, carbon monoxide and carbon (soot).
Are alkenes important?
Among the most important and most abundant organic chemicals produced worldwide are the two simple alkenes, ethylene and propylene. They are used as the starting materials to synthesize numerous valuable compounds.
Why alkane is called paraffin?
Alkanes are called paraffins because they have a little affinity towards a general reagent. In other words, alkanes are inert substances. They undergo reactions under drastic conditions.
What are alkenes and alkynes?
An alkene is a hydrocarbon with one or more carbon-carbon double covalent bonds. An alkyne is a hydrocarbon with one or more carbon-carbon triple covalent bonds.
Why ethene is an alkene?
The second subset of hydrocarbons is called the alkenes. Their names all end in –ene for example ethene. Alkenes all contain a carbon to carbon double bond which makes them more reactive than the alkanes.
Is methane an alkene?
A hydrocarbon that contains no double bonds is called an alkane, or hydrocarbon containing only single bonds. Methane and ethane are both alkanes.
Where are alkenes used?
What are alkenes used for? In manufacturing, alkenes find many different applications. They are used in the synthesis of alcohols, plastics, lacquers, detergents, and fuels as starting materials. For the chemical industry, the most important alkenes are ethene, propene, and 1,3-butadiene.
How do you write an alkene?
The Basic Rules for Naming Alkenes
- Find the longest carbon chain that contains both carbons of the double bond.
- Start numbering from the end of the parent chain which gives the lowest possible number to the double bond. ...
- Place the location number of the double bond directly before the parent name.
What is alkane nomenclature?
Alkanes can be described by the general formula C nH 2n +2. An alkyl group is formed by removing one hydrogen from the alkane chain and is described by the formula C nH 2n +1. The removal of this hydrogen results in a stem change from -ane to -yl.



