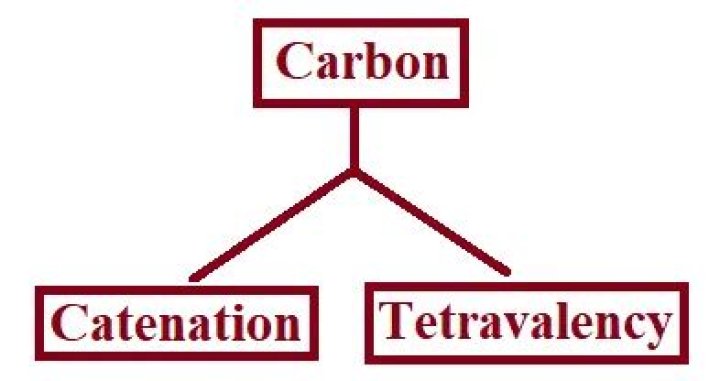
What is catenation and Tetravalency Class 10?

William Rodriguez
Published Jan 22, 2026
Catenation: It is the ability to form bonds with other atoms of carbon. Tetravalency: With the valency of four, carbon is capable of bonding with four other atoms.
What is catenation in 10th class?
Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon.
How can you define Tetravalency and catenation?
The key difference between catenation and tetravalency is that catenation includes the binding of atoms of the same chemical element to form chain or ring structures, whereas tetravalency refers to the ability to form four covalent bonds.
What is catenation and Tetravalency of carbon?
(a) Catenation: Carbon has the unique ability to form bonds with other atoms of carbon, giving rise to large molecules. This property is called catenation. (b) Tetravalency: Carbon has a valency of four, so it is capable of bonding with four other atoms of carbon or atoms of some other monovalent element.
What is a Tetravalency?
tetravalence in British English
1. the condition of having a valency of four. 2. the condition of having four valencies. the tetravalence of carbon.
43 related questions foundWhat is Covalency?
the number of covalent bonds that a particular atom can make with other atoms in forming a molecule.
What is meant by catenation?
catenation, chemical linkage into chains of atoms of the same element, occurring only among the atoms of an element that has a valence of at least two and that forms relatively strong bonds with itself.
What is the process of catenation?
In chemistry, catenation is the bonding of atoms of the same element into a series, called a chain. A chain or a ring shape may be open if its ends are not bonded to each other (an open-chain compound), or closed if they are bonded in a ring (a cyclic compound).
What is valence of hydrogen?
Hydrogen has a valence of 1, because one more electron will give a hydrogen atom an inert-gas structure. Carbon can form four bonds, because four more electrons give it the neon electronic structure.
What is catenation and isomerism?
Isomerism : The phenomenon of possessing same molecular formula but different properties by the compounds is known as isomerism. The compounds that exhibit isomerism are called isomers. Catenation : If an element forms bonds between its own atoms to give big molecule we call this property as catenation property.
What is saturated class 10?
Compounds formed by the linking of carbon by single bonds in between them are called saturated compounds. These compounds have hydrogen atoms that fill all of the other bonding orbitals of the carbon atoms. For example, Alkanes are saturated compounds. It is a series of saturated compounds.
What are the types of catenation?
The most common examples of catenation or elements that exhibit catenation are:
- Carbon.
- Silicon.
- Sulfur.
- Boron.
What is catenation 11th?
- Catenation is the binding of an element to itself through covalent bonds to form ring or chain molecules. For example, Carbon is the most common element that exhibits the property of catenation to maximum extent. It is capable of forming long hydrocarbon chains and rings like benzene.
What is meant by catenation name two?
(a) Catenation is the unique property exhibited by the carbon atoms. It is the ability of one carbon atom to combine with its other atoms to form long chains and give rise to a large number of carbon compounds. Carbon and silicon are the two elements which exhibit the property of catenation.
How do you pronounce catenation?
Phonetic spelling of catenation
- cate-na-tion.
- c-at-en-a-tion. Laurie Boyle.
- kat-n-ey-shuh n. Gregory Brekke.
- cate-na-tion. Ona Ullrich.
What is nitrogen valency?
So, the valency of nitrogen is 3.
What is lithium valency?
Lithium – Atomic number of lithium is 3. Its electronic configuration is 2,1. So, it needs to lose one electron to attain stability and get an electronic configuration like noble gas Helium. Thus, its valency is 1.
What is chain in chemistry?
referred to as the “chain,” and the atoms between which the chemical bonding takes place are said to make up the “backbone” of the chain. In most cases polymers are made up of carbon backbones—that is, chains of carbon (C) atoms linked together by single (C―C) or double (C=C)…
What is home eluga series?
A group of carbon compound having same general formula and same functional group is called homologous series. The members of homologous series are called homologue.
What is meant by catenation power?
Solution : Carbon has a unique ability to form strong covalent bonds with other carbon atoms, this result in formation of big molecules . This property of carbon is called catenation power.
What is Silicons reactivity?
Silicon, like carbon, is relatively inactive at ordinary temperatures; but when heated it reacts vigorously with the halogens (fluorine, chlorine, bromine, and iodine) to form halides and with certain metals to form silicides.
How do you find the covalence of a compound?
Covalency is the number of bonds an atom forms within a molecule. To determine the covalency, you draw the Lewis structure of the molecule and count the number of shared electron pairs.
What is co valency with example?
When an element shares electrons with other atoms of the same or different elements to acquire stable electronic configuration, it is called covalency. If an atom shares 1 electron, its covalency is equal to 1. If it can share 2 electrons, its covalency is 2. Covalency of Fluorine – Fluorine has 7 valence electrons.
What is Covalency of Aluminium?
The maximum covalency of Aluminium is 6.