What is meant by catenation Class 10?

William Rodriguez
Published Jan 12, 2026
Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon.
What is the meant by catenation?
catenation, chemical linkage into chains of atoms of the same element, occurring only among the atoms of an element that has a valence of at least two and that forms relatively strong bonds with itself.
What is catenation and Tetravalency Class 10?
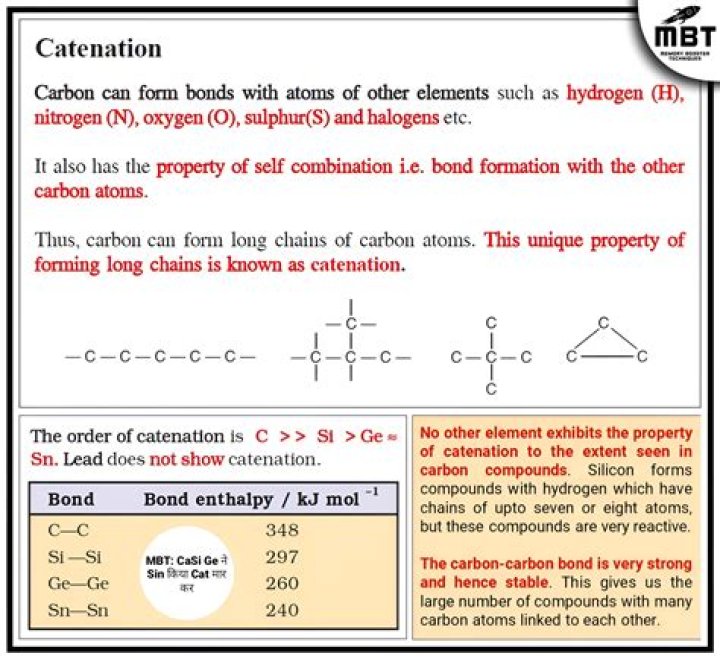
Catenation: It is the ability to form bonds with other atoms of carbon. Tetravalency: With the valency of four, carbon is capable of bonding with four other atoms.
What is catenation give two examples?
Catenation Definition: Catenation is the binding of an element to itself through covalent bonds to form chain or ring molecules. Examples: Carbon is the most common element that exhibits catenation. It can form long hydrocarbon chains and rings like benzene.
What is meant by catenation name two elements which show the property of catenation Class 10?
(a) The property of self combination of carbon atoms to form long chains is called catenation. Two elements which exhibit the property of catenation is Carbon and Silicon.
35 related questions foundWhat is meant by catenation power?
Solution : Carbon has a unique ability to form strong covalent bonds with other carbon atoms, this result in formation of big molecules . This property of carbon is called catenation power.
What is meant by catenation name one element other than carbon which exhibit the property of catenation?
Solutions : Answered on 13th Jan, 2021. (a) Catenation is the property of self-combination of carbon atoms to form a long chain. Carbon and silicon exhibit the property of catenation.
What is Heterocatenation and catenation?
As nouns the difference between catenation and heterocatenation. is that catenation is catenation (all senses) while heterocatenation is (chemistry) the catenation of two or more different elements or groups.
What is catenation Byjus?
Carbon atoms have a unique property of linking together in straight chains, branched chains or rings of various sizes and forming a large number of organic compounds. This property is known as catenation. Chemistry. Frank Chemistry - II.
What is catenation Brainly?
Brainly User. Answer: Catenation is the binding of an element to itself through covalent bonds to form chain or ring molecules. Examples: Carbon is the most common element that exhibits catenation. It can form long hydrocarbon chains and rings like benzene.
What is catenation and isomerism?
Isomerism : The phenomenon of possessing same molecular formula but different properties by the compounds is known as isomerism. The compounds that exhibit isomerism are called isomers. Catenation : If an element forms bonds between its own atoms to give big molecule we call this property as catenation property.
What is catenation Ncert?
Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon.
What is the process of catenation?
In chemistry, catenation is the bonding of atoms of the same element into a series, called a chain. A chain or a ring shape may be open if its ends are not bonded to each other (an open-chain compound), or closed if they are bonded in a ring (a cyclic compound).
What are the types of catenation?
As a result, a large number of organic compounds are due to the property of catenation of carbon elements. Three types of chains can be formed when carbon atoms combine. As shown below, there are three types of chains: straight chains branched chains, and closed chains or ring type chains.
What are isomers examples?
Isomers are molecules that have the same molecular formula, but have a different arrangement of the atoms in space. e.g. 1-pentene and 2-pentene are isomers.
How do you pronounce catenation?
Phonetic spelling of catenation
- cate-na-tion.
- c-at-en-a-tion. Laurie Boyle.
- kat-n-ey-shuh n. Gregory Brekke.
- cate-na-tion. Ona Ullrich.
What is catenation 9th?
Answer: Catenation is the binding of an element to itself or with other elements through covalent bonds to form an open chain or closed chain compounds. Carbon is the most common element which undergoes catenation and forms long-chain compounds.
What is catenation 11th?
- Catenation is the binding of an element to itself through covalent bonds to form ring or chain molecules. For example, Carbon is the most common element that exhibits the property of catenation to maximum extent. It is capable of forming long hydrocarbon chains and rings like benzene.
What are Carboranes with examples?
Carboranes have been prepared by many routes, the most common being addition of alkynyl reagents to boron hydride clusters to form dicarbon carboranes. For example, the high-temperature reaction of pentaborane(9) with acetylene affords several closo-carboranes as well as other products: nido-B5H9 + C2H2.
What is meant by catenation property of carbon?
Catenation is the ability of carbon to form long chains. In fact, carbon atoms are unique because of catenation—they are unique among all of the other atoms found in nature. Now, carbon chains are formed because they form bonds, or tetravalent bonds, with other carbon atoms.
What do you mean by catenation Class 8?
Answer: Catenation can be defined as the self-linking of atoms of an element to form chains and rings.
What is catenation name the element that shows maximum Carnation Why?
Carbon exhibits the property of catenation due to strong C-C bond.
Which of the following element forms catenation only upto seven or eight atoms?
Like carbon, silicon is another element which has the ability to form long chains. Since, silicon also shows properties similar to the properties of carbon atom. Hence after carbon atom, silicon atom has the ability to show catenation upto 7 or 8 atoms.
What are the isomers of hexane?
There are five isomers of hexane: n-hexane (nC6), 2-methylpentane (2MP), 3-methylpentane (3MP), 2,3-dimethylbutane (23DMB), and 2,2-dimethylbutane (22DMB) (Figure 1).
What are saturated hydrocarbons?
A Saturated hydrocarbon is a hydrocarbon in which all the carbon-carbon bonds are single bonds. A hydrocarbon is an organic compound whose only constituents are carbon and hydrogen.



