What is nucleophilic substitution reaction with example?

Daniel Moore
Published Jan 23, 2026
An example of nucleophilic substitution is the hydrolysis of an alkyl bromide, R-Br under basic conditions, where the attacking nucleophile is OH− and the leaving group is Br−. Nucleophilic substitution reactions are common in organic chemistry. Nucleophiles often attack a saturated aliphatic carbon.An example of nucleophilic substitution is the hydrolysis of an alkyl bromide, R-Br under basic conditions, where the attacking nucleophile is OH− and the leaving group The physical manifestation of leaving group ability is the rate at which a reaction takes place. Good leaving groups give fast reactions. By transition state theory, this implies that reactions involving good leaving groups have low activation barriers leading to relatively stable transition states. › wiki › Leaving_group
What is a nucleophilic substitution reaction?
Nucleophilic substitution reactions are a class of reactions in which an electron rich nucleophile attacks a positively charged electrophile to replace a leaving group. For alginate reactions, the most reactive nucleophile is the C6 carboxylate group.
What is substitution reaction explain with example?
A substitution reaction is also called a single displacement reaction, single replacement reaction, or single substitution reaction. Examples: CH3Cl reacted with a hydroxy ion (OH-) will produce CH3OH and chlorine. This substitution reaction replaces the chlorine atom on the original molecule with the hydroxy ion.
What is nucleophilic substitution class 12?
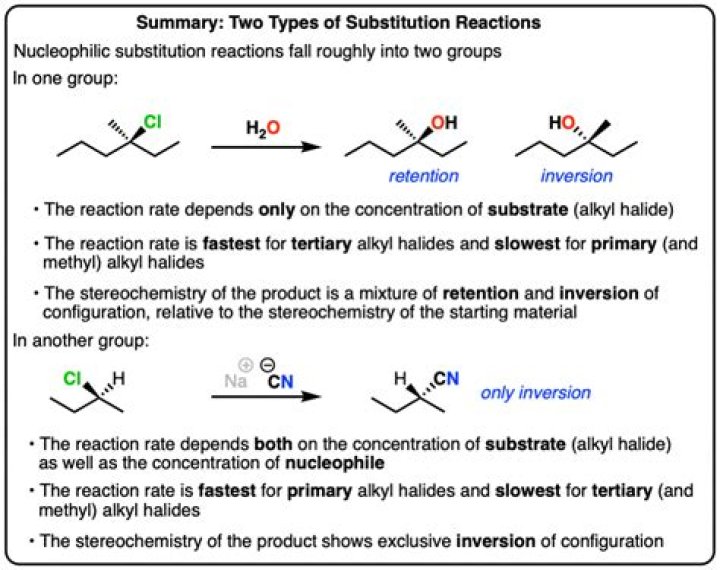
Nucleophilic substitution is a reaction in which a leaving group is replaced by an electron rich compound. It occurs in two ways: SN1 and SN2. SN1 reactions are unimolecular in rate of reaction and have a stepwise mechanism, while Sn2 reactions are bimolecular in rate of reaction and have a concerted mechanism.
What is nucleophilic and electrophilic substitution reaction?
Electrophilic substitutions involve displacement of a functional group by an electrophile (generally a hydrogen atom). Electrophiles are species that are attracted to electrons. Nucleophilic substitutions involve attack of a positively charged (or partially positively charged) atom or group by a nucleophile.
29 related questions foundWhat are electrophiles and nucleophiles with example?
Electrophiles are electron deficient species and can accept an electron pair from electron rich species. Examples include carbocations and carbonyl compounds. A nucleophile is electron rich species and donates electron pairs to electron deficient species. Examples include carbanions, water , ammonia, cyanide ion etc.
What are nucleophiles give two examples?
Examples of nucleophiles are the halogen anions (I-, Cl-, Br-), the hydroxide ion (OH-), the cyanide ion (CN-), ammonia (NH3), and water. Compare electrophile.
What is nucleophilic substitution reaction Class 11?
Nucleophilic substitution reaction is a class of organic reactions where one nucleophile replaces another. It is very similar to the normal displacement reactions which we see in chemistry, where, a more reactive element replaces a less reactive element from its salt solution.
What is substitution reaction with Example Class 10?
The conversion of methane to Carbon tetrachloride is a substitution reaction as hydrogen atoms are replaced by chlorine atoms.
How many types of nucleophilic substitution reactions are there?
It simply represents the Substitution of a Nucleophile for the Leaving Group. There are 2 types of Nucleophilic Substitution Reactions: SN1 & SN2, each having its own mechanism. The SN2 reaction is usually learned first.
What are the 3 main types of substitution reaction?
1. Nucleophilic Substitution
- SN1 reaction.
- SN2 reaction.
- Nucleophilic aliphatic substitution.
- Nucleophilic aromatic substitution.
- Nucleophilic acyl substitution.
Why is it called nucleophilic substitution?
Technically, this is known as an SN2 reaction. S stands for substitution, N for nucleophilic, and the 2 is because the initial stage of the reaction involves two species - the bromoethane and the Nu- ion. If your syllabus doesn't refer to SN2 reactions by name, you can just call it nucleophilic substitution.
What is substitution reaction of carbon compounds give examples?
Substitution reaction of carbon compounds
Alkanes undergo substitution reactions. Example: Chlorination of methane in presence of sunlight gives a mixture of products like methyl chloride, methylene chloride, chloroform and carbon tetrachloride.
What is substitution reaction give Example Class 11?
Substitution reaction:
-Example of substitution reaction is halogenation of alkanes. In this reaction methane reacts with chlorine to form methyl chloride and hydrochloric acid.
What is substitution class 10 Ncert?
CBSE class 10 Science - Carbon and its Compounds - Substitution reaction is a type of reaction in which one atom or a group of atoms takes the place of another atom. Saturated hydrocarbons are less reactive because they have their nobel gas attained, however they can react with chlorine in sunlight at a high speed.
What are electrophiles give an example class 11?
Answer: Electrophiles are atoms or molecules known to be electron deficient and that carry a partial or fully positive charge and will seek an electron pair to form a covalent bond . An example of an electrophile is a Lewis Acid. Other examples include Br+, Cl+, and CH3+.
Which of the following is an example of electrophiles?
Examples of electrophiles are hydronium ion (H3O+, from Brønsted acids), boron trifluoride (BF3), aluminum chloride (AlCl3), and the halogen molecules fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2).
What are electrophiles explain electrophile substitution reaction with the help of example?
Electrophilic Aromatic Substitution Reaction
In electrophilic aromatic substitution reactions, an atom attached to an aromatic ring is replaced with an electrophile. Examples of such reactions include aromatic nitrations, aromatic sulphonation, and Friedel-Crafts reactions.
What are nucleophiles give examples Class 11?
A nucleophile is a species that gives an electron pair to form a covalent bond. Nucleophiles are usually negatively charged or is neutral with a lone couple of donatable electrons. These are electron-rich species. Examples are ammonia, cyanide ion, etc.
What are nucleophiles 11?
Nucleophiles are nucleus loving chemical species. Since the nucleus of any atom is positively charged, therefore, nucleophiles must be electron rich chemical species containing at least one lone pair of electrons.
What are electrophiles and nucleophiles Shaalaa com?
An electrophile is an electron-deficient species. A nucleophile is an electron-rich species.
What is substitution reaction carbon and compounds 10?
A Substitution reaction is one in which an atom or a group of atoms(functional group) in the compound are replaced by another atom (or group of atoms). Substitution reactions are single displacement reactions. Alkanes, which have only single bonds between their carbon atoms, are saturated hydrocarbons.
What is homologous series explain with an example?
Answer : A homologous series is a series of carbon compounds that have different numbers of carbon atoms but contain the same functional group. For example, methane, ethane, propane, butane, etc. are all part of the alkane homologous series. The general formula of this series is CnH2n+2.
What is combustion addition and substitution reaction?
(ii) Substitution reaction:- A reaction in which one atom of a molecule is replaced by another atom(or group of atoms) is called a substitution reaction. (iii) Combustion reaction:- Carbon in all its allotropic forms, burns in oxygen to give carbon dioxide and water along with the release of heat and light.
What is E2 reaction?
E2 Definition. The E2 reaction - A Nucleophilic Elimination reaction in which the Rate Determining Step involves 2 components. -E2 reactions are bimolecular, with simultaneous bond-making and bond breaking steps. -E2 reactions do not proceed through an intermediate.