What is the chemical formula of hydrazine?

James Craig
Published Jan 07, 2026
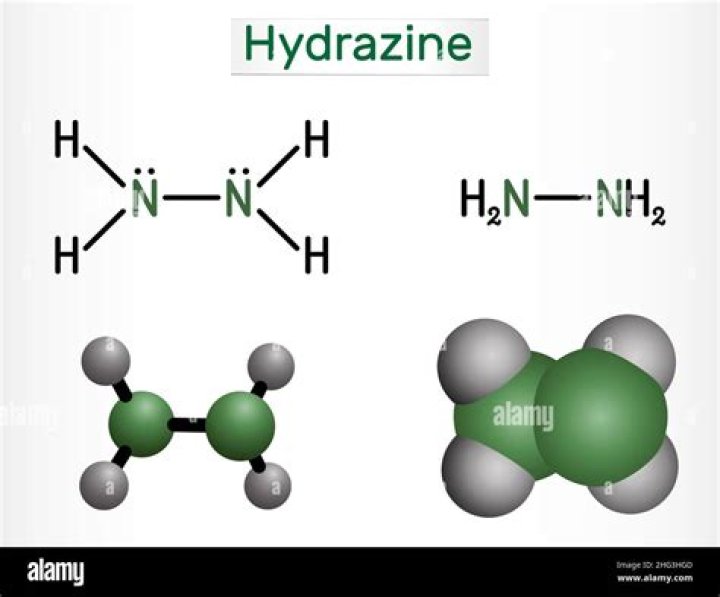
Hydrazine is an inorganic compound with the chemical formula N₂H₄. It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly toxic unless handled in solution as, for example, hydrazine hydrate.
What is the chemical name for hydrazine?
Hydrazine, N2H4, is a chemical compound synthesized by Theodor Curtius in 1889. Also known as diazane, hydrozine has a density of 1.01g/mL, a melting point of 274K and a boiling point of 384K. Hydrazine is a chemical compound with the formula of N2H4.
What is the chemical equation for the production of hydrazine?
hydrazine preparation
2NH3 + NaOCl → N2H4 + NaCl + H2O This reaction is known to occur in two main steps.
What is hydrazine also known as?
Hydrazine is also called as Diamine or Diazane or Nitrogen hydride and is a strong base. It is an azane and dangerously unstable. Each subunit of H2N-N is pyramidal and the N−N bond distance is about 1.45 Å. Diamine in its anhydrous form, is a colourless, fuming oily liquid which smells like ammonia.
What is the chemical formula of hydrogen peroxide?
Hydrogen peroxide is a chemical compound with the formula H2O2. In its pure form, it is a very pale blue liquid, slightly more viscous than water.
17 related questions foundHow is hydrazine acid prepared?
Preparation: The commercial production of hydrazine is by the Raschig process, in which sodium hypochlorite solution is treated with excess ammonia to form a chloramine intermediate, which then gives the final hydrazine product along with hydrochloric acid.
What is the molecular formula of formaldehyde?
Synonyms: methanal, Methyl aldehyde, Methylene glycol, Methylene oxide, Formalin - H2C(OH)2, Formol, Carbonyl hydride. Formaldehyde is a naturally occurring organic compound with the formula CH2O (H-CHO).
What is hydrogen oxide?
hydrogen oxide (uncountable) (inorganic chemistry) The simple systematic name for water, H2O.
How is chloramine made?
Chloramines are formed during a reaction between chlorine ) and ammonia (NH3). Chloramines are amines which contain at least one chlorine atom, which is directly bond to nitrogen atoms (N). Inorganic chloramines are formed when dissolved chlorine and ammonia react.
How is Monochloramine made?
Monochloramine is formed by a reaction between chlorine and ammonia. It is commonly used because it results in reduced levels of certain disinfection byproducts (DBPs) while still maintaining adequate disinfection. This is important because some studies have indicated that high levels of DBPs may cause health problems.
Why is n20 called laughing gas?
Nitrous Oxide is also called laughing gas or happy gas due to its intoxicating effects when inhaled. It was initially discovered around 1772 by the English scientist and clergyman Joseph Priestley (who was also famous for being the first to isolate other important gases such as oxygen and carbon dioxide among others).
What is laughing gas Class 12?
Nitrous oxide (N2O), also known as dinitrogen monoxide, laughing gas or nitrous, is one of several nitrogen oxides, a colourless gas with a fun, sweet smell and taste that, when inhaled, induces insensitivity to pain accompanied by mild hysteria, often laughter.