What is the importance of catenation?

Noah Mitchell
Published Jan 18, 2026
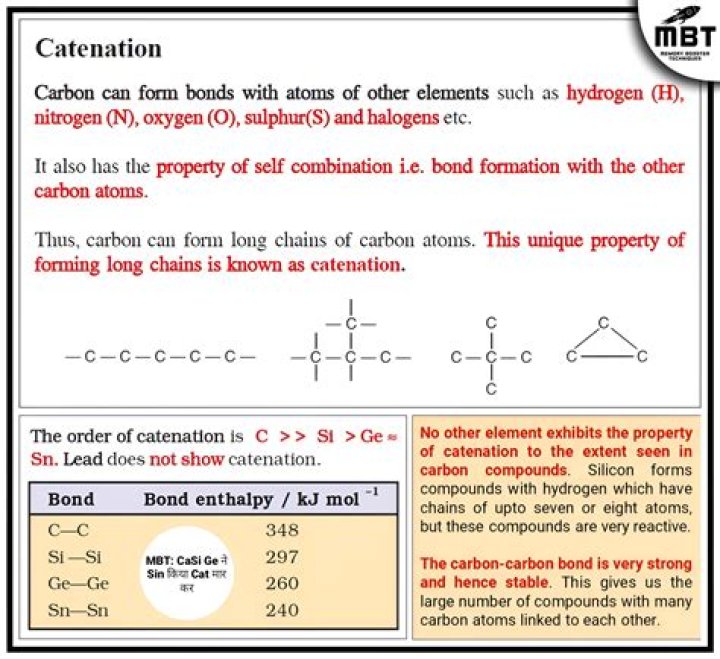
Catenation is the ability of carbon to form long chains. In fact, carbon atoms are unique because of catenation—they are unique among all of the other atoms found in nature. Now, carbon chains are formed because they form bonds, or tetravalent bonds, with other carbon atoms.
Why is catenation important?
Catenation is the linkage of atoms of the same element into longer chains. Catenation occurs most readily in carbon, which forms covalent bonds with other carbon atoms to form longer chains and structures. This is the reason for the presence of the vast number of organic compounds in nature. Was this answer helpful?
For which element catenation is most important?
Carbon is most well known for its properties of catenation, with organic chemistry essentially being the study of catenated carbon structures (and known as catenae).
What is catenation in short answer?
Catenation can be defined as the self-linking of atoms of an element to form chains and rings.
What do you mean by catenation Class 10?
Catenation is the ability of an atom to form bonds with other atoms of the same element.
26 related questions foundWhat is a catenation give example?
Catenation Definition: Catenation is the binding of an element to itself through covalent bonds to form chain or ring molecules. Examples: Carbon is the most common element that exhibits catenation.
What is catenation power?
Solution : Carbon has a unique ability to form strong covalent bonds with other carbon atoms, this result in formation of big molecules . This property of carbon is called catenation power.
What is catenation Class 9?
Answer: Catenation can be defined as the self-linking of atoms of an element to form chains and rings.
What are the properties of catenation?
The property of catenation is predominant in Carbon. The capability of atoms forming a long chain by bonding with each other called catenation. It is the linkage of atoms of the same element to form long chains.
How do you pronounce catenation?
Phonetic spelling of catenation
- cate-na-tion.
- c-at-en-a-tion. Laurie Boyle.
- kat-n-ey-shuh n. Gregory Brekke.
- cate-na-tion. Ona Ullrich.
What is meant by catenation capacity?
The large number of organic compounds is due to the ability of carbon atom to form long chains with other carbon atoms through the sharing of electrons. This unique property of carbon is known as catenation.
What is the effect of catenation in terms of the formation of chemicals?
Catenation of chemical bonds leads to the formation of inorganic polymers. However, inorganic polymers are mostly solids in the form of crystals. Typical inorganic polymers are diamond, graphite, silicates, and other solids in which all atoms are connected by covalent bonds.
What is meant by catenation name two?
(a) Catenation is the unique property exhibited by the carbon atoms. It is the ability of one carbon atom to combine with its other atoms to form long chains and give rise to a large number of carbon compounds. Carbon and silicon are the two elements which exhibit the property of catenation.
What do you understand by catenation and Tetravalency?
Catenation: It is the ability to form bonds with other atoms of carbon. Tetravalency: With the valency of four, carbon is capable of bonding with four other atoms.
Which has the highest catenation power?
Explanation: Carbon has the highest degree of catenation because: High C-C bond energy.
Which has maximum catenation power?
Carbon shows catenation to a maximum extent because of its electronic configuration and tendency to form strong covalent bonds. The bond energy of C-C bond is greater than N-N bond and thus catenation in nitrogen is restricted to 2 or 3 nitrogen atoms.
Why is carbon used in catenation?
Reason: Carbon exhibits catenation much more than silicon or any other element due to its smaller size which makes the C−C bonds strong while the Si−Si bonds are comparatively weaker due to its large size and therefore, Si does not have enough bond strength to hold together large molecules.
What is the catenation class11?
- Catenation is the binding of an element to itself through covalent bonds to form ring or chain molecules. For example, Carbon is the most common element that exhibits the property of catenation to maximum extent. It is capable of forming long hydrocarbon chains and rings like benzene.
What is Covalency Class 11?
When an element shares electrons with other atoms of the same or different elements to acquire stable electronic configuration, it is called covalency. If an atom shares 1 electron, its covalency is equal to 1. If it can share 2 electrons, its covalency is 2.
What is catenation Byjus?
Carbon atoms have a unique property of linking together in straight chains, branched chains or rings of various sizes and forming a large number of organic compounds. This property is known as catenation. Chemistry. Frank Chemistry - II.
What is catenation Brainly?
Brainly User. Answer: Catenation is the binding of an element to itself through covalent bonds to form chain or ring molecules. Examples: Carbon is the most common element that exhibits catenation. It can form long hydrocarbon chains and rings like benzene.
What is the catenation power of carbon?
The unique property of carbon atom to form covalent bonds with other carbon atoms giving rise to extremely big molecules is called catenation power. The covalent bond between two carbon atoms is strong and stable. This is the reason carbon is bestowed with catenation power.
Does oxygen have catenation power?
Solution : Oxygen has only a little catenation power because the `O-O` bond is very weak.
What do you understand by the term catenation and isomerism?
Therefore, the compounds that exhibit isomerism are known as isomers. what is catenation. Catenation Definition: Catenation is the binding of an element to itself through covalent bonds to form chain or ring molecules. Examples: Carbon is the most common element that exhibits catenation.
What is catenation name the element which shows catenation?
(a) The property of self combination of carbon atoms to form long chains is called catenation. Two elements which exhibit the property of catenation is Carbon and Silicon.