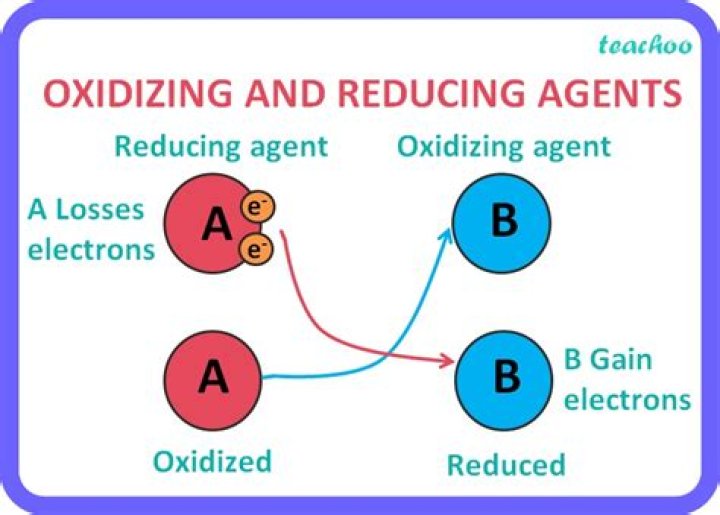
What is the weakest oxidizing agent?

Sarah Smith
Published Jan 23, 2026
Answer and Explanation: H2O2 H 2 O 2 is a weakest oxidising agent because it can act as a reducing agent also.Answer and Explanation: H2O2 H 2 O 2 Answer and Explanation: Hydrogen peroxide, a solution that's used as a bleaching agent and antibacterial/microbial agent, is a weak acid with a pH of 6.2. › is-hydrogen-peroxide-an-acid-or-base
How do you know which oxidizing agent is strongest to weakest?
The higher the electronegativity the greater the pull an oxidizing agent has for electrons. The higher the pull for electrons the stronger the oxidizing agent. So the element with the highest electronegativity is the strongest oxidizing agent.
Which is the weakest reduction agent?
Hydrogen has most positive E0 value. Thus hydrogen has least tendency to donate electron and is the weakest reducing agent.
What does a weak oxidizing agent mean?
Weak oxidizing agents will react less vigorously than a strong oxidizing agent, but can still participate in reactions that generate heat and possibly gaseous products which can pressurize a closed container, and which may go on to participate in further reactions.
What is the strongest and weakest reducing agent?
One will get Na, Cr, Cu and Cl-; Na being the strongest reducing agent and Cl- being the weakest one. A few good common reducing agents include active metals such as potassium, calcium, barium, sodium and magnesium and also, compounds that contain the H- ion, those being NaH, LiAlH4 and CaH2.
42 related questions foundWhat is a strong oxidizing agent?
Strong oxidizing agents are typically compounds with elements in high oxidation states or with high electronegativity, which gain electrons in the redox reaction (Figure 1). Examples of strong oxidizers include hydrogen peroxide, permanganate, and osmium tetroxide.
Which is best oxidizing agent?
Elemental fluorine, for example, is the strongest common oxidizing agent.
What is mild oxidizing agent?
Mild oxidising agents are those which do not completely oxidise the alcohol to carboxylic acids.
Is H2O2 an oxidizing agent?
Oxidation with Hydrogen Peroxide
Hydrogen peroxide has the chemical formula H2O2 and is an oxidizing agent that is similar to oxygen in effect but is significantly stronger. The oxidizing activity of hydrogen peroxide results from the presence of the extra oxygen atom compared with the structure of water.
Which is the weakest reducing agent in Group 1?
Lithium is the weakest reducing agent among alkali metals.
Which of the following is weakest reducing agent in Group 15?
Ammonia is the weakest reducing agent as well as the weakest base among Group 15 hydrides.
What's the strongest reducing agent?
Lithium, having the largest negative value of electrode potential, is the strongest reducing agent.
Which is the strongest reducing agent in Group 15?
(iii) BiH3 is the strongest reducing agent amongst all the hydrides of group 15 elements.
Is co2 an oxidizing agent?
The abundant availability, non-toxic, economic and mild oxidizing properties of CO2 has resulted in immense interest in its use as an oxidant in several reactions, such as the oxidative coupling of CH4 and the oxidative dehydrogenation of alkanes and alkyl aromatics.
Is kmno4 an oxidizing agent?
Potassium permanganate is a very strong oxidizing agent and can, therefore, be used as an oxidant in a wide spectrum of chemical reactions.
Is h20 oxidized or reduced?
Hydrogen peroxide (H2O2) In this molecule the oxidation number for oxygen is –1. This is halfway between O2(0) and H2O(–2), and so hydrogen peroxide can either be reduced or oxidized.
Why is PCC a weak oxidizing agent?
Explanation: PCC is an oxidizing agent. It converts alcohols to carbonyls, but is not strong enough to convert a primary alcohol into a carboxylic acid. It only converts primary alcohols to aldehydes, and secondary alcohols to ketones.
What is mild reducing agent?
Iodides– The salts of Iodides are said to be mild reducing agents. They react with oxygen to give out iodine. These also possess various antioxidant properties.
Is k2cr2o7 a strong oxidizing agent?
Potassium dichromate is a powerful oxidizing-agent, especially in an acidic medium. This chemical compound is commonly used to oxidize alcohols.
Why Pb 4 is a strong oxidising agent?
In contrast, the inert pair effect is' more prominent in Pb than in Sn. Therefore, +2 oxidation state of Pb is more stable than its +4 oxidation state. In other words, Pb4+ can easily lose two electrons to form Pb2+ and hence Pb4+ acts as an oxidising agent.
Which has the highest oxidizing power?
Fluorine is the most effective oxidizer, having the largest positive electrode potential. Fluorine is thought to be the most powerful elemental oxidizing agent.
Is mg a strong reducing agent?
Yes, magnesium is a stronger reducing agent than zinc. Metals have the tendency to lose electrons.
Which is the strongest reducing agent in group 1?
Lithium, which has a high ionisation energy value in alkali metals, serves as the solution's main reducing agent. It is, however, observed that Li is the strongest reducing agent in solution among alkali metals as Li's E value is maximum.
Why is BiH3 strongest reducing agent?
Since,the BiH3 is the least stable (because the size of central atom is greatest & therefore its tendency to form stable covalent bond with small hydrogen atom decreases,as a result the bond strength decreases) in this series,hence the reducing character increases.
Why is d3 the strongest reducing agent among all the hydrides of group 15 elements?
The stability of these hydride decreases while moving down the group. The nitrogen hydride is more stable and bismuth hydride is least stable. Due to the least stability of bismuth hydride, it is the strongest reducing agent among all the hydrides of group \[15\] elements.