What structure type is diamond?

James Craig
Published Jan 20, 2026
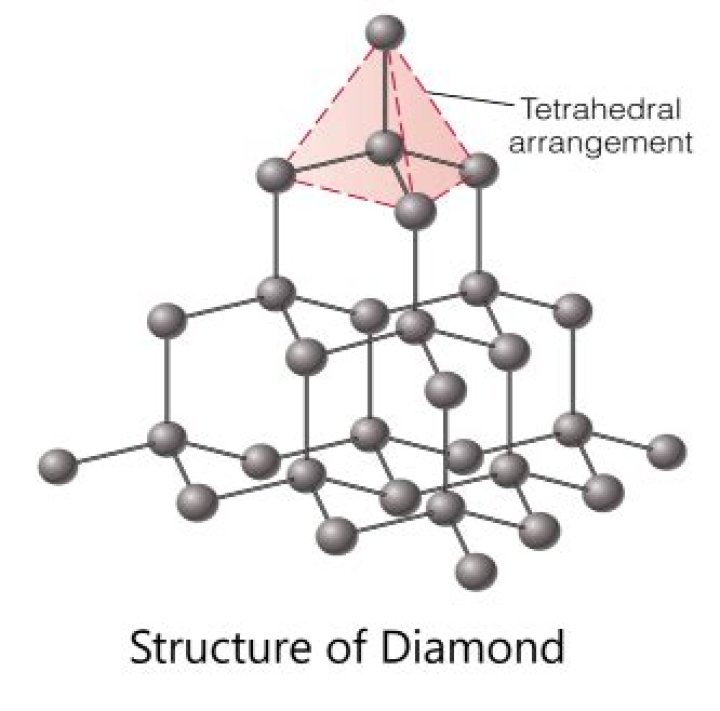
Diamond is a giant covalent structure in which: each carbon atom is joined to four other carbon atoms by strong covalent bonds. the carbon atoms form a regular tetrahedral network structure. there are no free electrons.
Is diamond A BCC?
The diamond structure is thus fcc with a basis containing two identical atoms. is at the center, and its four NNs are at the corners of the cube (or vice versa). Each atom forms four bonds with its NNs. Atoms in diamond-type crystals form covalent bonding.
Is diamond a simple molecule or extended structure?
Examples of simple molecules could include ammonia and methanol. Examples of extended structures could include sodium chloride or diamonds. Examples of molecular-level models could include drawings, 3D ball and stick structures, or computer representations showing different molecules with different types of atoms.]
Is diamond a 3d structure?
Because the diamond structure forms a distance-preserving subset of the four-dimensional integer lattice, it is a partial cube. Yet another coordinatization of the diamond cubic involves the removal of some of the edges from a three-dimensional grid graph.
Is diamond covalent or ionic?
Diamond is a giant covalent structure in which: each carbon atom is joined to four other carbon atoms by strong covalent bonds. the carbon atoms form a regular tetrahedral network structure. there are no free electrons.
21 related questions foundIs diamond a compound?
The quick answer is: Diamond is a pure element, carbon; gold is a pure element, gold; and rust is a compound, Iron Oxide, of iron and Oxygen. Diamond is pure elemental carbon, compressed to its crystal form, under extreme heat and pressure deep within the Earth.
Is diamond tetrahedral or octahedral?
In two of these structures, diamond has only tetrahedral symmetry and in the two others the full or octahedral symmetry of the cubic system. The selection rules require that the fundamental vibration of the diamond lattice having a frequency 1332 cm.
What is the structure of diamond and graphite?
Diamond is a type of carbon that has its atoms arranged in a diamond cubic crystal structure. Another solid form of carbon known as graphite is the chemically stable form of carbon at ambient temperature and pressure, but diamond almost never transforms to it.
Are diamonds octahedral?
The most common shape for rough gem quality diamond is the octahedron, which looks like two pyramids back to back. Crystals that are almost perfect in shape and transparency are called glassies. Without any one of these factors, diamond might be just another mineral.
What is fullerene structure?
Fullerenes are molecules composed entirely of carbon with spherical (buckyballs), ellipsoid, tubular (nanotubes) or a combination shape (nanobuds). They consist of hexagonal and pentagonal (sometimes also heptagonal) rings, with the latter necessary for the curvature of the molecule.
Is diamond homogeneous or heterogeneous?
It's a heterogeneous mixture. Diamond is made of just one element: carbon. Each carbon atom in diamond is connected to four other carbon atoms, in a crystal that extends on and on. There are other forms of pure carbon where the atoms are bonded differently, notably charcoal and graphite.
Is diamond a substance or mixture?
Diamond is neither a compound nor a mixture. Diamond is an element since it purely consists of a single type of atom, which is Carbon (C). No matter how many times you break it down, it will stay as Carbon and it cannot produce other elements.
Is diamond a metal?
Diamond is not a metal in anyway its just an allotrope of carbon. It does not show any physical properties or chemical properties of metals like electrical conductivity, malleability, ductility, reaction with acids or salts etc.
Why is a diamond a mixture?
In the case of diamonds, most of the material is carbon. Within the carbon, you'll find the impurities in trace amounts. These impurities (various elements or other compounds) are not chemically bonded to the carbon. Since the impurities are not bonded to the carbon, the diamond is considered to be a mixture.
Why is diamond not an element?
No. Diamond is not an element. It is a name for a gemstone, a particular occurence of an allotropic form of carbon that may or may not contain other elements in addition to carbon.
Why diamond is an element?
Originally Answered: Why is a diamond an element? Because carbon is the only substance in its composition. C2, C60, C1000000 are all compositions containing atoms of carbon arranged in different ways. Carbon is the only substance in these compositions, so they are all various forms or allotropes of the element carbon.
How do you separate diamonds?
The heavy minerals, including diamonds, are separated from waste material based on their density differences in a dense media separation process. In this process, ore is mixed into a ferrosilicon slurry, then fed to a dense media cyclone, where the heavy minerals are separated from the waste.
Is a diamond shape?
A diamond is a quadrilateral, a 2-dimensional flat figure that has four closed, straight sides. But a diamond is also categorized as rhombus, because it has four equal sides and its opposite angles are equal. And, because its opposite sides are parallel, it's also considered to be a parallelogram.
What is diamonds scientific name?
Dia. Strunz classification. 1.CB.10a. Dana classification. 1.3.6.1.
What is the biggest diamond in the world?
1. The Cullinan Diamond. The world's biggest diamond is the Cullinan Diamond, discovered in the Premier Mine in South Africa in 1905. Named for the owner of the mine, Thomas Cullinan, the Cullinan Diamond was a massive 3,106 carats in its uncut state: that's 1-1/3 pounds!
What is structure of graphite?
Graphite has a giant covalent structure in which: each carbon atom is joined to three other carbon atoms by covalent bonds. the carbon atoms form layers with a hexagonal arrangement of atoms. the layers have weak forces between them. each carbon atom has one non-bonded outer electron, which becomes delocalised.
What's the structure of graphene?
Graphene is a two-dimensional carbon allotrope. It is composed of carbon atoms positioned in a hexagonal design, which can be said to resemble a chicken wire. A single layer of carbon atoms arranged in such a honeycomb structure forms a single graphene sheet.



