What type of reaction is bromination of alkenes?

James Craig
Published Jan 18, 2026
[Worth noting: bromination of alkenes is technically an oxidation reaction, because each carbon goes from being bound to another carbon (0) to bromine (–1). The oxidation state of each carbon in ethene is +2; the oxidation state of each carbon in dibromoethane is +1. ]
What type of reaction is bromination?
The bromination of benzene is an example of an electrophilic aromatic substitution reaction. In this reaction, the electrophile (bromine) forms a sigma bond to the benzene ring, yielding an intermediate.
What type of reaction is bromination of alkanes?
Alkanes undergo a substitution reaction with halogens in the presence of light. For instance, in ultraviolet light , methane reacts with halogen molecules such as chlorine and bromine. This reaction is a substitution reaction because one of the hydrogen atoms from the methane is replaced by a bromine atom.
What type of reaction is halogenation of alkenes?
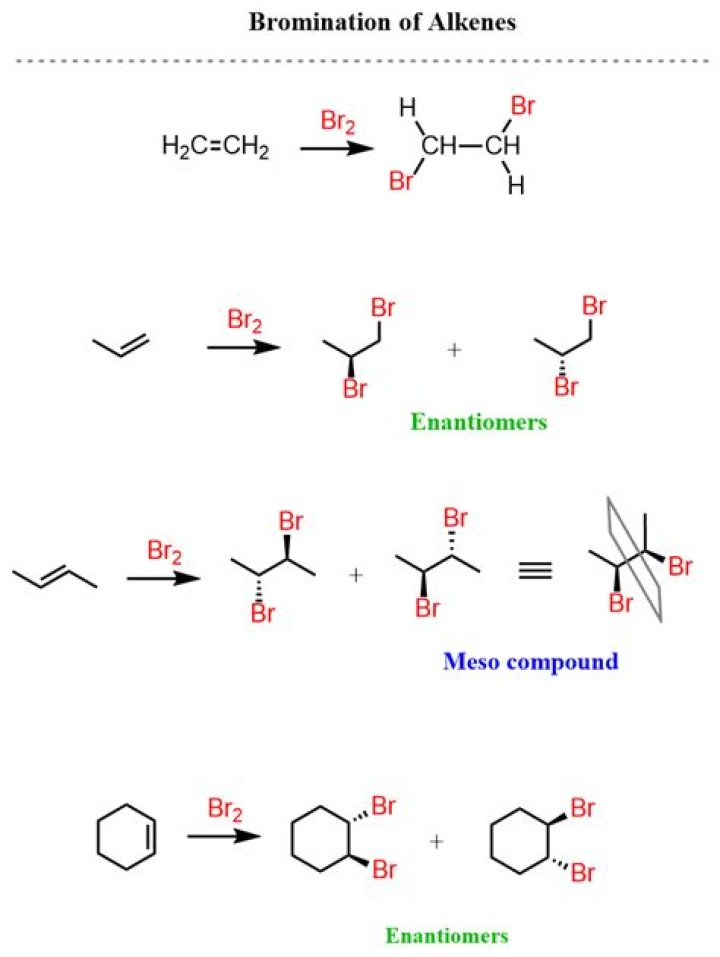
Reaction Overview: The alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as Cl2 or Br2 is added to a molecule after breaking the carbon to carbon double bond. The halides add to neighboring carbons from opposite faces of the molecule.
What type of reaction is bromination and chlorination?
In chlorination, the reaction is exothermic, and the transition state resembles the reactants. According to Hammond's postulate, we could say that this transition state is “early”. In bromination, the reaction is endothermic, and the transition state resembles the products.
19 related questions foundWhat reaction do alkenes undergo?
Like the alkanes , the alkenes undergo combustion . However, alkenes are less likely to combust completely , so they tend to burn in air with a smoky flame due to incomplete combustion .
What are the chemical reactions of alkenes?
Alkenes react with cold concentrated sulfuric acid to form alkyl hydrogen sulfate ester. This product is formed by the addition of hydrogen of acid to one carbon of alkene double bond and bisulfate ion to the other. On diluting the reaction mixture and warming it up, sulfate ester is hydrolyzed to form alcohol.
Why does alkene react with bromine?
The electrophilic addition of bromine to ethene
Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The double bond breaks, and a bromine atom becomes attached to each carbon.
Is bromination of alkenes stereospecific?
Bromination of alkenes is stereospecific because the geometry of the starting alkene determines which diastereoisomer is obtained as the product. Bromination of Z– and E-2-butene in acetic acid produces a single diastereoisomer in each case, both of which are different from each other.
Why bromination of alkene is anti?
The stereospecificity of bromine addition can be explained by considering the anti-addition or trans-addition. alkene to form a flat carbocation. Then the bromide ion would attack the bottom face of the alkene. Thus anti-addition to cis-2 butene leads to the formation of an enantiomer.
Do alkenes undergo combustion reactions?
Alkanes and alkenes both undergo complete combustion , but only alkenes can undergo addition reactions .
Does alkenes undergo substitution reaction?
Alkanes undergo only substitution reactions but alkenes and alkynes undergo both substitution and addition reactions.
Is bromination of an alkene oxidation or reduction?
[Worth noting: bromination of alkenes is technically an oxidation reaction, because each carbon goes from being bound to another carbon (0) to bromine (–1).
What does a bromination reaction do?
Bromination: Any reaction or process in which bromine (and no other elements) are introduced into a molecule. Bromination of an alkene by electrophilic addition of Br2. Bromination of a benzene ring by electrophilic aromatic substitution.
What intermediates are involved in the bromination of alkene?
Answer - 1) Reaction of bromine – Reaction mechanism – Involved intermediate of reaction mechanism for the bromination of an alkene is bromonium ion...
Is bromination anti or syn?
In anti addition, two substituents are added to opposite sides (or faces) of a double bond or triple bond, once again resulting in a decrease in bond order and increase in number of substituents. The classical example of this is bromination (any halogenation) of alkenes.
Is bromine addition stereospecific or stereoselective?
The addition of bromine to alkenes is a stereospecific reaction.
What is the name of the reaction when alkenes react with HCl?
We said earlier that the reaction of HCl and HBr (among others) with alkenes is “regioselective”. In this post we give several examples of these regioselective reactions and trace them back to the observations of a Russian chemist in the 1880's, Vladimir Markovnikov.
What are stereoselective and stereospecific reactions?
Stereospecific and stereoselective reactions are two types of reaction that can be found in organic chemistry. The main difference between stereospecific and stereoselective reactions is that a stereospecific reaction gives one specific product whereas stereoselective reaction gives multiple products.
How do alkenes react with bromine water?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot.
Is the bromine test an addition reaction?
The bromine test is used to test for an unsaturated carbon carbon bond, such as an alkene or alkyne. The test uses a type of chemical reaction called addition, where a reactant, here bromine, is added to an organic compound to break a double or triple bond.
What is bromination of alkenes used for?
Bromination of Alkenes Gives anti Products
It's a family of reactions which proceed through 1) attack of an alkene upon an acid, forming a free carbocation, and 2) attack of a nucleophile upon the carbocation.
What are the 4 reactions of alkenes?
Addition reactions involving alkenes and alkynes include hydrogenation, halogenation, and hydrohalogenation. Alkenes and alkynes are useful reagents in polymer synthesis—an important industrial application.
What is the most common reaction of alkenes?
The most common chemical reaction undergone by alkenes is the addition reaction. This reaction involves the transformation of a carbon-carbon double bond into a single bond via the addition of other functional groups.
Why do alkenes undergo addition reaction?
Alkenes are unsaturated molecules, which means they do not have all the hydrogen they could have. This is because there is at least one double bond between carbons. This is a stable structure, but not the most stable, so when certain compounds or elements are added, like fluorine, they undergo an addition reaction.