What type of reaction is electrophilic addition?

Noah Mitchell
Published Jan 22, 2026
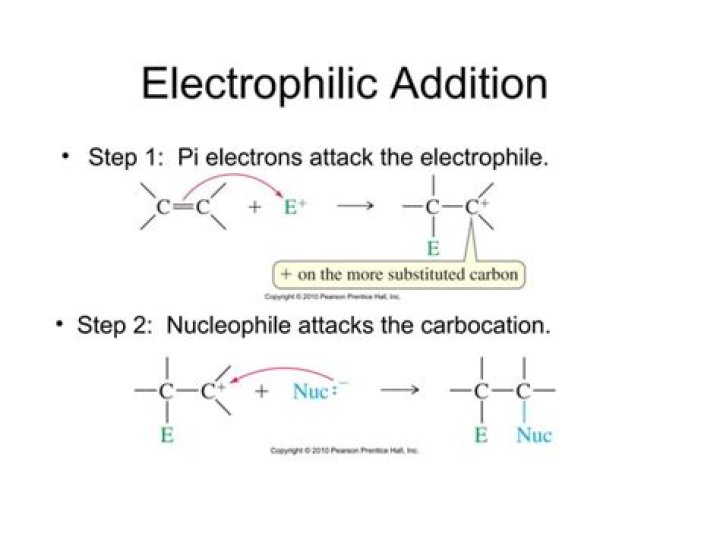
In organic chemistry, an electrophilic addition reaction is an addition reaction where a chemical compound containing a double or triple bond has a π bond broken, with the formation of two new σ bonds.
Which reaction is electrophilic addition?
An electrophilic addition reaction is a reaction in which a substrate is initially attacked by an electrophile, and the overall result is the addition of one or more relatively simple molecules across a multiple bond.
What are the 4 types of addition reactions?
Different Types of Addition Reaction
- Nucleophilic addition reaction.
- Electrophilic addition reaction.
- Free radical addition reaction.
Is electrophilic addition E1?
The reverse of electrophilic addition is called E1 elimination.
Why is it called electrophilic addition reaction?
An electrophilic addition reaction is an addition reaction which happens because what we think of as the "important" molecule is attacked by an electrophile. The "important" molecule has a region of high electron density which is attacked by something carrying some degree of positive charge.
36 related questions foundIs hydrogenation an electrophilic addition reaction?
Is hydrogenation electrophilic addition? There is another alkene reaction, hydrogenation, which merits notice but is not related to the mechanism of electrophilic addition. The addition of molecular hydrogen (H2) to the alkene double bond is termed hydrogenation. It transforms a simple alkene into an alkane.
What is an electrophilic addition reaction chegg?
Electrophilic addition reaction is a type of addition reaction in which a pi bond is broken to form two new sigma bonds. In the compound of ethene, there is a double bond between the two carbon atoms, which are formed by the sharing of two pairs of electrons.
Is electrophilic addition stereospecific?
29 models in this collection. Use getProperty "modelInfo" or getProperty "auxiliaryInfo" to inspect them. Bromination of alkenes is stereospecific because the geometry of the starting alkene determines which diastereoisomer is obtained as the product.
What do you mean by electrophilic substitution reaction?
Electrophilic substitution reactions are chemical reactions in which an electrophile displaces a functional group in a compound, which is typically, but not aromatic substitution reactions are characteristic of aromatic compounds and are common ways of introducing functional groups into benzene rings.
Is halogenation an electrophilic addition reaction?
(X represents the halogens bromine or chlorine, and in this case, a solvent could be CH2Cl2 or CCl4). The product is a vicinal dihalide. This type of reaction is a halogenation and an electrophilic addition.
What is a type of addition reaction?
There are two main types of polar addition reactions: electrophilic addition and nucleophilic addition. Two non-polar addition reactions exist as well, called free-radical addition and cycloadditions. Addition reactions are also encountered in polymerizations and called addition polymerization.
What is E2 reaction?
E2 Definition. The E2 reaction - A Nucleophilic Elimination reaction in which the Rate Determining Step involves 2 components. -E2 reactions are bimolecular, with simultaneous bond-making and bond breaking steps. -E2 reactions do not proceed through an intermediate.
What is addition reaction class11?
In organic chemistry, an addition reaction is an organic reaction in which two or more molecules combine to generate a bigger one (the adduct). Molecules with carbon—hetero double bonds, such as carbonyl (C=O) or imine (C=N) groups, can be added because they have double-bond character as well.
What is the difference between electrophilic addition reaction and electrophilic substitution reaction?
When an electrophile replace an atom from an compound the reaction is called electrophilic substitution reaction. while when an electrophile directly add to the compound the reaction is called electrophilic addition .
Why do alkenes prefer electrophilic addition reaction?
Alkenes are rich source of loosely held pi (π) electrons due to which they show electrophilic addition reaction . Electrophilic addition reaction of alkenes are accompanied by large energy changes so these are energetically favourable than of electrophilic substitution reactions.
How many types of electrophilic substitution reactions are there?
There are two types of electrophilic substitution reactions undergone by organic compounds. They are: Electrophilic Aromatic Substitution Reactions. Electrophilic Aliphatic Substitution Reactions.
What is nucleophilic and electrophilic substitution reaction?
Electrophilic substitutions involve displacement of a functional group by an electrophile (generally a hydrogen atom). Electrophiles are species that are attracted to electrons. Nucleophilic substitutions involve attack of a positively charged (or partially positively charged) atom or group by a nucleophile.
What is an example of electrophilic substitution?
substitution reaction
This reaction is known as electrophilic substitution. Examples of electrophilic species are the hydronium ion (H3O+), the hydrogen halides (HCl, HBr, HI), the nitronium ion (NO2+), and sulfur trioxide (SO3).
What are stereoselective and stereospecific reactions?
Stereospecific and stereoselective reactions are two types of reaction that can be found in organic chemistry. The main difference between stereospecific and stereoselective reactions is that a stereospecific reaction gives one specific product whereas stereoselective reaction gives multiple products.
What is the mechanism for electrophilic addition to alkenes?
Electrophilic addition to alkenes starts with the pi electrons attacking an electrophile, forming a carbocation on the most stable carbon. A nucleophile then attacks the carbocation to form the product. There are many different kinds of such addition, including: Hydroxylation.
What is addition reaction 10th?
CBSE class 10 Science - Carbon and its Compounds - Addition reaction is a reaction in which one molecule combines with another molecule to form a larger molecule with no other products. Carbon compounds use addition reaction to convert Unsaturated hydrocarbon into saturated hydrocarbon.
What are the different types of alkene addition reaction?
Addition reactions involving alkenes and alkynes include hydrogenation, halogenation, and hydrohalogenation.
What is Wurtz reaction Class 11?
Wurtz's reaction is an organic chemical coupling reaction wherein sodium metal is reacted with two alkyl halides in the environment provided by a solution of dry ether in order to form a higher alkane along with a compound containing sodium and the halogen.
Which reaction is example of E2 reaction?
Examples of E2 Reactions
Alkyl halides undergo elimination to produce alkenes. Alcohols undergo elimination to produce alkenes. Notice that E2 elimination of an alcohol uses acid, not base.