Why is an alkene an electrophile?

Emily Ross
Published Jan 06, 2026
Why do alkenes undergo electrophilic addition reactions? Alkenes are doubly bound and sp2 hybridized, which can be donated to an electrophile, such as electrophilic addition, by the electrons in the side-to-side overlap of p orbitals that allows the pi bond.
Why is alkene electrophilic?
In this section, the electrophilic addition reaction will be discussed in general to provide a better understanding of subsequent alkene reactions. As discussed in Section 6-5, the double bond in alkenes is electron rich due to the prescience of 4 electrons instead of the two in a single bond.
Is an alkene a nucleophile or electrophile?
Yes, alkenes are nucleophiles. The π bond is localized above and below the C-C σ bond. These π elecrons are relatively far from the nuclei and are loosely bound. An electrophile can attract those electrons and pull them away to form a new bond.
Can an alkene act as an electrophile?
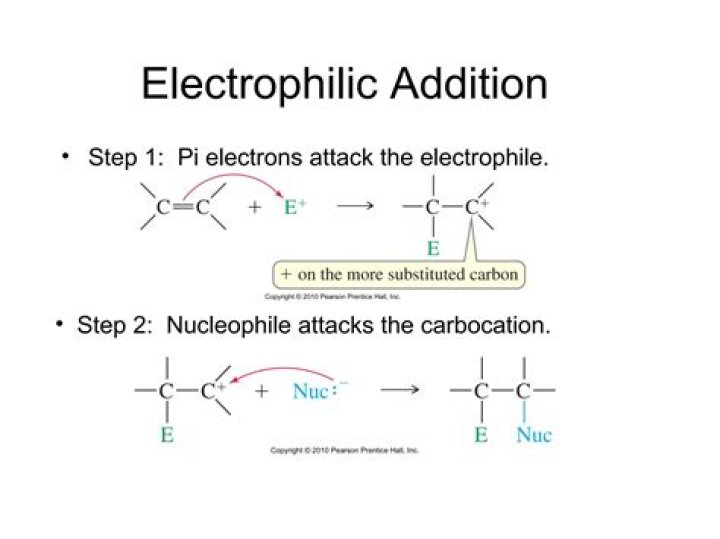
Electrophilic addition is probably the most common reaction of alkenes. Consider the electrophilic addition of H-Br to but-2-ene: The alkene abstracts a proton from the HBr, and a carbocation and bromide ion are generated. The bromide ion quickly attacks the cationic center and yields the final product.
Why is double bond electrophilic?
In the functional group of an alkene - the carbon-carbon double bond -- the most readily available electrons are those in the pi bond. They are farther from the nuclei than the electrons in a sigma bond, so they are more readily attracted to an electrophile if one approaches.
17 related questions foundWhy is alkene a nucleophile?
Alkene has a double bond, meaning it wants to donate an electron. Meaning it acts as the nucleophile. Therefore it cannot react with a reagent that wants to donate an electron. You cannot react two compounds that want to both give an electron.
Why is electrophilic addition called electrophilic addition?
An electrophilic addition reaction is an addition reaction which happens because what we think of as the "important" molecule is attacked by an electrophile. The "important" molecule has a region of high electron density which is attacked by something carrying some degree of positive charge.
Why is a double bond a nucleophile?
Overall, double bonds can easily donate lone pair electrons to act like a nucleophile (nucleus-loving, electron rich, a Lewis acid). During an electrophilic addition reactions double bonds donate lone pair electrons to an electrophile (Electron-loving, electron poor, a Lewis base).
Why do alkenes react?
Alkenes are relatively stable compounds, but are more reactive than alkanes because of the reactivity of the carbon–carbon π-bond. Most reactions of alkenes involve additions to this π bond, forming new single bonds. The carbon-carbon double bond in alkenes such as ethene react with concentrated sulfuric acid.
Why is it important to identify the reaction of the alkenes?
The high reactivity make alkenes an important type of organic compounds, and they can be used to the synthesis a wide variety of other compounds, such as halide, alcohol, ethers, alkanes. The most common type of reaction for alkene is the addition reaction to C=C double bond.
Is h2so4 a nucleophile?
Sulphuric acid acts as a nucleophile. A nucleophile is usually charged negatively with a lone couple of donatable electrons. An anion is always a better nucleophile than that of a neutral molecule, so a better nucleophile is always the conjugate base.
Can alkenes undergo nucleophilic substitution?
Alkenes undergo electrophilic addition whereas aldehydes and ketones undergo nucleophilic addition.
What is hydrogenation of alkenes?
An example of an alkene addition reaction is a process called hydrogenation.In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane.
Why do alkenes prefer to undergo electrophilic addition rather than nucleophilic?
Answer. *Alkenes are rich source of loosely held pi electrons, due to which they show electrophilic addition reaction. Electrophilic addition reactions of alkenes are accompanied by large energy changes so these are energetically favourable than that of electrophilic substitution reactions.
Why do alkenes show geometrical isomerism?
Explanation: Alkenes can also show geometrical isomerism. This occurs due to the restriction to rotation about the C = C bond (the double bond is rigid). ... * For the fact that alkenes show both structural and geometrical isomerism, a particular alkene has more isomers than it corresponding alkane.
Why are alkenes more reactive than alkanes?
Alkenes are unsaturated, meaning they contain a double bond . This bond is why the alkenes are more reactive than the alkanes .
Why alkenes are described as being unsaturated hydrocarbons?
The alkenes are unsaturated hydrocarbons: hydrocarbons , because they are compounds containing hydrogen and carbon only. unsaturated, because they contain a C=C double bond, which means that they have two fewer hydrogen atoms than the corresponding alkane.
What does h2so4 do to alkenes?
Alkenes react with concentrated sulfuric acid in the cold to produce alkyl hydrogensulphates. For example, ethene reacts to give ethyl hydrogensulphate. The structure of the product molecule is sometimes written as CH3CH2HSO4, b ut the version in the equation is better because it shows how all the atoms are linked up.
Why is a double bond more reactive?
Double bonds are often found in alkenes and ring structures, where the double bond gives more stability due to resonance. They are more reactive than single bonds because they are more electron rich.
What are reactions of alkenes?
Alkenes react with cold concentrated sulfuric acid to form alkyl hydrogen sulfate ester. This product is formed by the addition of hydrogen of acid to one carbon of alkene double bond and bisulfate ion to the other. On diluting the reaction mixture and warming it up, sulfate ester is hydrolyzed to form alcohol.
What are the main reactions of alkenes?
- Reactions of Alkenes and Alkynes. Alkenes and alkynes are generally more reactive than alkanes due to the electron density available in their pi bonds. ...
- Addition Reactions. ...
- Cycloaddition. ...
- Oxidation. ...
- Hydrogenation. ...
- Halogenation. ...
- Hydrohalogenation. ...
- Hydration.
What is electrophilic and nucleophilic addition?
A nucleophilic addition reaction has a nucleophile being added up. This nucleophile provides or donates electrons on the place of its addition. While an electrophilic addition reaction has an electrophile, which is an electron deficient species that accepts electrons.
What is the mechanism for electrophilic addition in alkenes by heterolytic fission?
Alkenes react with a mechanism called electrophilic addition. This involves the breaking of the weak pi bond and a molecule being added in an addition reaction. All four of these reactions occur using this mechanism. Advantages compared to fermentation: Pure product formed, continuous process.
What is the difference between electrophilic addition and nucleophilic addition?
Electrophilic addition is where the group being added accepts an electron pair while nucleophilic addition is where the group being added donates an electron pair.



