Will phosphoric acid eat metal?

Rachel Ellis
Published Jan 08, 2026
Strong acids will dissolve rust, but they will also dissolve paint, finishes, and sometimes even the metal itself. Hydrochloric acid (which is also called muriatic acid in its diluted form), as well as phosphoric acid and sulfuric acid may be used in rust removal formulas using strong acids.Strong acids will dissolve rust, but they will also dissolve paint, finishes, and sometimes even the metal itself. Hydrochloric acid (which is also called muriatic acid in its diluted form), as well as phosphoric acid and sulfuric acid may be used in rust removal formulas using strong acids.
Will phosphoric acid dissolve metal?
Most acids will etch metal whether they cause rust or remove it. Phosphoric acid (H3PO4) is one type of acid which removes rust by converting it (iron III oxide) to a form which can be dissolved in water.
Is phosphoric acid safe on steel?
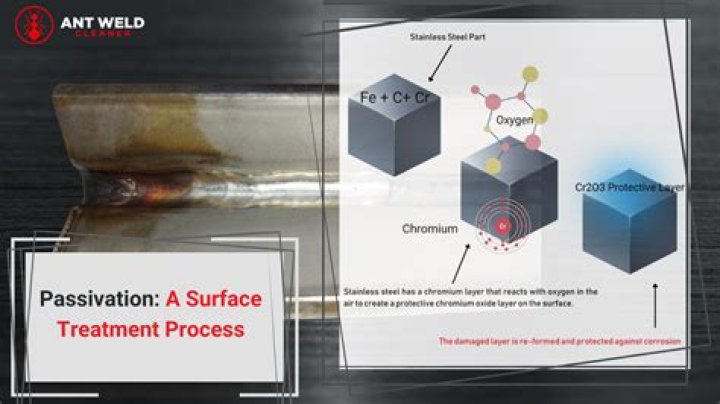
Phosphoric acid is a strong chemical that can remove tough stains from the surface of stainless steel. This substance is particularly useful in removing rust stains caused by carbon steel contamination, according to the British Stainless Steel Association.
What acid can eat through metal?
Hydrochloric acid is so strong that it can eat through metal, something you can witness first hand in the school chemistry lab.
Does phosphoric acid harm aluminum?
Phosphoric acid, in liquid or jelly form, is not recommended for use on painted steel, chrome, or even stainless steel. And these acids will quickly dissolve aluminum components.
43 related questions foundCan phosphoric acid dissolve iron?
Phosphoric acid effectively removes the rust because it does not actually dissolve it. It bonds with the rust to create iron III oxide and water.
Does phosphoric acid remove galvanizing?
If the galvanized is showing signs of red rust, then it is best to clean it with a phosphoric acid cleaner to remove that first. If there are significant amounts of it, it may not be possible to recover this product. The white corrosion product can be removed with a weaker organic acid such as acetic acid.
Does hydrofluoric acid dissolve metal?
Hydrofluoric acid is corrosive stuff. Hydrofluoric acid attacks the silicon oxide in most types of glass. It also dissolves many metals (not nickel or its alloys, gold, platinum, or silver), and most plastics.
Can acid eat through glass?
In short, acid can dissolve glass. However, only a few acids, mainly acids containing the element fluorine, are corrosive enough to properly dissolve glass. An example of this is hydrofluoric acid, which is able to react with the compound silicon dioxide found in glass, thus dissolving it.
Can hydrofluoric acid melt steel?
'Breaking Bad' Profile: Hydrofluoric Acid
Hydrofluoric acid won't eat through plastic. It will, however, dissolve metal, rock, glass, ceramic. So there's that.
How long does it take for phosphoric acid to remove rust?
Rust removers that use phosphoric acid include Rust-Oleum Rust Dissolver Gel, Boeshield Rust Free and Krud Kutter “The Must for Rust” Rust Remover and Inhibitor Gel. Instructions differ slightly. Rust Free should be left on for a minute or less. The others treat light rust in 10 to 30 minutes.
How long do you leave phosphoric acid on rust?
Usually, you'll have to wait for about five minutes.
What strength of phosphoric acid removes rust?
Rust Removal: Use Phosphoric Acid diluted to 20% (200ml/litre. Apply to surface for 5-30 minutes to remove light film. Soak very rusty items for days with periodic agitation. Rinse with cold water.
Will phosphoric acid stop rust?
Dilute to a 25-50% concentration with water and soak rusted metal parts. Over time, rust will remove and the phosphoric acid reacts with the metal to create a rust inhibiting barrier on the metal.
Does phosphoric acid convert rust?
Phosphoric acid converts iron oxide (rust) into an inert layer of iron phosphate, which is black in colour. This inert layer then acts as a barrier layer or protective coating. A rust converter eliminates the need to sandblast rust therefore less time and energy is involved in the process.
Can acid damage steel?
Acids. Strong acids destabilize the passive layer. When stainless steel is exposed to hydrochloric and sulfuric acids, general surface corrosion can occur.
Does acid dissolve gold?
Gold alloys, despite being harder than pure gold, can be dissolved in acids more easily than pure gold. Thus, the lower the karat, the more easily the gold alloy is dissolved. See the equations below, which show the results of adding concentrated nitric acid, HNO3, or aqua regia to different karats of gold.
Why can't acid eat through plastic?
It is capable of dissolving many materials, especially oxides. Hydrofluoric acid is usually stored in plastic containers due to its high reactivity. Plastic contains some contents which are considered as a resistance for the Hydrochloric acid, so hydrochloric acid does not dissolve plastic.
What is the deadliest acid?
The world's strongest superacid is fluoroantimonic acid, HSbF6. It is formed by mixing hydrogen fluoride (HF) and antimony pentafluoride (SbF5). Various mixtures produce the superacid, but mixing equal ratios of the two acids produces the strongest superacid known to man.
Why is hydrofluoric acid corrosive?
corrosive because of the H+ ions of the acid; and toxic because of the fluoride ions F– which, thanks to the destruction of the superficial layer of skin or eye, can penetrate deeply, chelate calcium and magnesium, and thus disrupt the biological balances.
What neutralizes hydrofluoric acid?
You can neutralize small spills (100 mL or less) by covering with magnesium sulfate (dry) and absorbing with spill control pads or other absorbent materials. Add sodium bicarbonate or magnesium oxide to any absorbent and place in a plastic container for disposal. Wash the spill site with a sodium bicarbonate solution.
Why is HF so reactive?
The difference in electronegativity means that the Hydrogen atom has little of the electron density and is easily removed or separated from the Florine creating a high concentration of free Hydrogen ions H+1 This makes Hydroflouric acids extremely reactive.
What neutralizes phosphoric acid?
The most common chemicals used to neutralize acids or bases are sodium hydroxide (50%) and sulfuric acid (98%). Sodium carbonate (soda ash), ammonium hydroxide, calcium hydroxide (lime), or magnesium hydroxide can also be used to raise the pH of an acidic liquid.
How do you neutralize phosphoric acid on steel?
Warning
- Clear the area of other people. ...
- Ventilate the area thoroughly. ...
- Sprinkle spills with either baking soda or powdered garden lime (both bases). ...
- Allow the base to sit on the acid, absorbing as it neutralizes. ...
- Sweep the sodden base and acid mixture up using a broom and dust pan.
Which acid is best for removing rust?
The most commonly used rust removing chemical is phosphoric acid. The solution creates a chemical reaction when applied to rust converting it to a water-soluble compound that can be scrubbed away quickly and easily.